Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhejiang Arts & Crafts Imp & Exp.co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjartspharm.com | |||
 | +86 571 87238903 | |||
 | jocelynpan@zjarts.com | |||
| Chemical distributor since 1992 | ||||
| chemBlink Standard supplier since 2024 | ||||
| IOLITEC Ionic Liquids Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.iolitec-usa.com | |||
 | +1 (205) 348-2831 | |||
 | +1 (205) 348-3510 | |||
 | info@iolitec-usa.com | |||
| Chemical manufacturer since 2002 | ||||
| Combi-Blocks, Inc. | USA | |||
|---|---|---|---|---|
 | www.combi-blocks.com | |||
 | +1 (858) 635-8950 | |||
 | +1 (858) 635-8991 | |||
 | sales@combi-blocks.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Ionic liquid |
|---|---|
| Name | 1-Butyl-3-methylimidazolium nitrate |
| Molecular Structure | 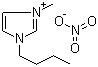 |
| Molecular Formula | C8H15N3O3 |
| Molecular Weight | 201.22 |
| CAS Registry Number | 179075-88-8 |
| EC Number | 629-008-8 |
| SMILES | CCCCN1C=C[N+](=C1)C.[N+](=O)([O-])[O-] |
| Hazard Symbols |   GHS03;GHS07 Danger Details GHS03;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H272-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P210-P220-P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Transport Information | UN 1479 | ||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
1-Butyl-3-methylimidazolium nitrate (BMIM NO₃) is a well-known ionic liquid with the chemical formula C₉H₁₁N₃O₃. This compound belongs to the broader class of ionic liquids, which have attracted attention for their unique properties and wide applications in various scientific and industrial fields. The development of BMIM NO₃ stems from the growing interest in ionic liquids, which began in the late 20th century. Researchers sought to create solvents with favorable properties such as low volatility, high thermal stability, and high ionic conductivity. The synthesis of BMIM NO₃ was designed to take advantage of these properties by combining the 1-butyl-3-methylimidazolium cation with the nitrate anion. The synthesis involves the reaction of 1-butyl-3-methylimidazolium chloride (BMIM Cl) with sodium nitrate (NaNO₃), in which BMIM Cl reacts with NaNO₃ to form BMIM NO₃ and NaCl. The process is usually carried out in a suitable solvent or directly in the ionic liquid phase, followed by purification to separate the ionic liquid. BMIM NO₃ is used in electrochemical applications due to its good ionic conductivity and stability. It can be used as an electrolyte in a variety of energy storage and conversion devices, including batteries and supercapacitors. Its low viscosity and high thermal stability improve the performance and efficiency of these devices, making it a valuable component in the field of energy technology. As a green solvent, BMIM NO₃ provides an environmentally friendly alternative to traditional organic solvents. It is non-volatile and recyclable, thus reducing the environmental impact of chemical processes. BMIM NO₃ is used in organic synthesis and extraction processes to improve reaction efficiency and selectivity while minimizing waste. In catalysis, BMIM NO₃ can be used as both a solvent and a co-catalyst. Its ability to dissolve a wide range of organic and inorganic compounds makes it a versatile medium for catalytic reactions. BMIM NO₃ is used in a variety of catalytic processes, both homogeneous and heterogeneous, to increase reaction rates and yields. BMIM NO₃ is very effective in separation processes, such as liquid-liquid extraction and ion exchange. Its unique solvation properties allow for the selective extraction and separation of specific compounds from mixtures. This application is particularly useful in drug purification and the recovery of valuable metals from ores. In materials science, BMIM NO₃ is used to synthesize and modify materials with tailored properties. It plays a role in the preparation of nanoparticles and advanced polymers. BMIM NO₃ can act as a solvent or stabilizer during the synthesis of nanomaterials, affecting their size, shape, and stability. References 2023. Tutorial for thermal analysis of ionic liquids. Journal of Thermal Analysis and Calorimetry. DOI: 10.1007/s10973-023-12439-z 2020. Applications of Ionic Liquids in Chemical Reactions. Nanotechnology-Based Industrial Applications of Ionic Liquids. DOI: 10.1007/978-3-030-44995-7_15 2021. Structural and Conformational Analysis of 1-Butyl-3-methylimidazolium Nitrate. Journal of Chemical Crystallography. DOI: 10.1007/s10870-021-00899-w |
| Market Analysis Reports |