Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhejiang Hisun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hisunpharm.com | |||
 | +86 13651899693 | |||
 | yiwen.sun@hisunpharm.com | |||
| Chemical manufacturer since 1956 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Synthetic anti-infective drugs >> Antiviral drugs |
|---|---|
| Name | Maribavir |
| Synonyms | 1263W94; BW 1263W94; GW 257406X; 5,6-Dichloro-N-(1-methylethyl)-1-beta-L-ribofuranosyl-1H-benzimidazol-2-amine |
| Molecular Structure | 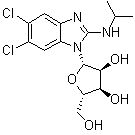 |
| Molecular Formula | C15H19Cl2N3O4 |
| Molecular Weight | 376.23 |
| CAS Registry Number | 176161-24-3 |
| EC Number | 891-156-6 |
| SMILES | CC(C)NC1=NC2=CC(=C(C=C2N1[C@@H]3[C@H]([C@H]([C@@H](O3)CO)O)O)Cl)Cl |
| Solubility | Practically insoluble (0.019 g/L) (25 °C), Calc.*, 50 mg/mL (DMSO) (Expl.) |
|---|---|
| Density | 1.67±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.703, Calc.* |
| Boiling Point | 611.0±65.0 °C (760 mmHg), Calc.* |
| Flash Point | 323.3±34.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS05;GHS07;GHS08 Dander Details GHS05;GHS07;GHS08 Dander Details |
|---|---|
| Risk Statements | H302-H318-H317 Details |
| Safety Statements | P260-P264-P264+P265-P270-P280-P301+P317-P305+P354+P338-P308+P316-P317-P330-P405-P501 Details |
| SDS | Available |
|
Maribavir is a novel antiviral drug developed for the treatment of cytomegalovirus (CMV) infections, particularly in immunocompromised patients, such as those undergoing organ transplantation or receiving chemotherapy. CMV is a common virus that can cause severe complications in individuals with weakened immune systems. Maribavir represents a promising option for treating CMV, particularly in cases where the virus has become resistant to other antiviral therapies. The discovery of maribavir was part of a broader effort to develop targeted therapies for viral infections that are difficult to treat with existing drugs. CMV infections can lead to a range of health problems, including pneumonia, retinitis, and gastrointestinal disorders, especially in patients with compromised immune systems. Traditional antiviral agents like ganciclovir and valganciclovir have been used to manage CMV infections; however, the emergence of drug-resistant strains has limited their effectiveness. This has created an urgent need for new therapeutic options. Maribavir was developed by the biopharmaceutical company ViroPharma (now part of Vir Biotechnology) and works by inhibiting the viral enzyme pUL97, which is essential for the replication and maturation of the CMV virus. Unlike other antiviral drugs that target viral DNA synthesis, maribavir inhibits the phosphorylation of viral proteins, thereby preventing the virus from replicating. This mechanism of action offers a unique approach to treating CMV infections, particularly in cases where resistance to other antiviral drugs is a concern. In preclinical and clinical studies, maribavir has demonstrated strong efficacy in treating CMV infections. It has shown significant activity against CMV strains that are resistant to ganciclovir and valganciclovir, providing a valuable therapeutic option for patients who may not respond to these traditional treatments. The clinical trials for maribavir have also indicated that it has a favorable safety profile, with most side effects being mild to moderate in severity. Common side effects include diarrhea, fatigue, and headache, but these are generally well-tolerated by patients. Maribavir received breakthrough therapy designation from the U.S. Food and Drug Administration (FDA) for the treatment of CMV infections in immunocompromised patients. This designation accelerated its development and approval process. In 2021, maribavir was approved by the FDA for the treatment of adult and pediatric patients with refractory CMV infections or those who are resistant to existing therapies. The approval of maribavir represents a significant advancement in the management of CMV, offering a new option for patients who previously had limited treatment alternatives. In addition to its use in treating CMV infections in transplant recipients and other immunocompromised individuals, maribavir is being studied for its potential to treat other viral infections and conditions. Its unique mechanism of action and ability to target drug-resistant strains of CMV have positioned it as a key therapeutic candidate in the ongoing battle against viral infections. In conclusion, maribavir is a breakthrough antiviral therapy that offers a targeted and effective approach to treating cytomegalovirus infections, particularly in patients with compromised immune systems. Its approval has filled an important gap in the treatment of CMV, especially for those who are resistant to other antiviral agents. References 1999. Benzimidavir. 1263W94, BW 1263W94, GW 1263. Drugs in R & D, 2(5). DOI: 10.2165/00126839-199902050-00004 2024. Human cytomegalovirus: pathogenesis, prevention, and treatment. Molecular Biomedicine, 5. DOI: 10.1186/s43556-024-00226-7 2024. Maribavir treatment for resistant cytomegalovirus disseminated disease in kidney transplant recipients: A case-based scoping review of real life data in literature. Transplantation reviews (Orlando, Fla.), 38. DOI: 10.1016/j.trre.2024.100873 |
| Market Analysis Reports |