Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Omkar Specialty Chemicals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.omkarchemicals.com | |||
 | +91 (251) 269-7340 | |||
 | +91 (251) 269-7347 | |||
 | info@omkarchemicals.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Samrat Remedies Limited | India | |||
|---|---|---|---|---|
 | www.samratremedies.com | |||
 | +91 (22) 2610-2491 | |||
 | +91 (22) 2610-1982 | |||
 | contact@samratremedies.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| VIVAN Life Sciences Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.vivanls.com | |||
 | +91 (22) 2544-4584 | |||
 | +91 (22) 2544-4588 | |||
 | vivan@vivanls.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Haimen RuiYi Medical Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ruiyitech.cn | |||
 | +86 (21) 5471-1706 | |||
 | +86 (21) 5471-1707 | |||
 | sales@ruiyitech.com zyj3625@msn.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Joyochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.joyochem.com | |||
 | +86 13290333633 | |||
 | sales@joyochem.com | |||
 | QQ Chat | |||
 | WeChat: 13290333633 | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Jinan An-dm Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.an-dm.com | |||
 | +86 15550736712 | |||
 | andmchem@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Jinan Aery Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aerpharm.com | |||
 | +86 (531) 8126-3406 +86 15553134202 | |||
 | +86 (531) 5859-1634 | |||
 | sales@aerpharm.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Jinan Great Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemgreat.com | |||
 | +86 (531) 5866-8191 | |||
 | +86 (531) 8632-7119 | |||
 | sale001@chemgreat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Sai Chemie Inc. | India | |||
|---|---|---|---|---|
 | www.saichemie.com | |||
 | +91 (816) 935-6778 | |||
 | saichemicalinc@gmail.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Cfm Oskar Tropitzsch GmbH | Germany | |||
|---|---|---|---|---|
 | www.cfmot.de | |||
 | +49 (9231) 9619-0 | |||
 | +49 (9231) 9619-60 | |||
 | info@cfmot.de | |||
| Chemical distributor since 1985 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Chunghwa Chemical Synthesis & Biotech Co., Ltd. | Taiwan | |||
|---|---|---|---|---|
 | www.ccsb.com.tw | |||
 | +886 (2) 8684-3318 | |||
 | +886 (2) 8684-3202 | |||
 | ccsb@ccsb.com.tw | |||
| Chemical manufacturer since 1964 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| C D N Isotopes Inc. | Canada | |||
|---|---|---|---|---|
 | www.cdnisotopes.com | |||
 | +1 (800) 565-4696 (514) 697-6254 | |||
 | +1 (514) 697-6148 | |||
 | info@cdnisotopes.com | |||
| Chemical manufacturer since 1993 | ||||
| Classification | Chemical reagent >> Organic reagent >> Sulfone, sulfoxide compound |
|---|---|
| Name | Trimethylsulfoxonium iodide |
| Synonyms | Trimethylsulphoxonium iodide |
| Molecular Structure | 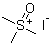 |
| Molecular Formula | C3H9IOS |
| Molecular Weight | 220.07 |
| CAS Registry Number | 1774-47-6 |
| EC Number | 217-204-2 |
| SMILES | C[S+](=O)(C)C.[I-] |
| Melting point | 208 - 212 °C (Decomposes) (Expl.) |
|---|---|
| Solubility | water 15 g/L (20 °C) (Expl.) |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H303-H315-H319-H320-H335-H401-H411 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Trimethylsulfoxonium iodide is an organosulfur compound that belongs to the class of sulfoxonium salts. It is a quaternary sulfoxonium salt in which a sulfur atom is bonded to three methyl groups and one oxygen atom, forming a positively charged sulfoxonium cation, paired with an iodide anion. The compound has been primarily investigated and applied as a reagent in organic synthesis, particularly in the formation of epoxides and other three-membered ring systems through ylide intermediates. The discovery of trimethylsulfoxonium iodide is linked to the broader development of sulfoxonium and sulfonium salts in the mid-20th century, as chemists explored the utility of onium compounds in stabilizing reactive intermediates. Trimethylsulfoxonium iodide is especially noted for its ability to generate dimethyloxosulfonium methylide upon treatment with strong bases such as sodium hydride. This sulfoxonium ylide is a highly reactive species that can participate in a variety of transformations, most notably the Corey–Chaykovsky reaction, which allows the conversion of carbonyl compounds into epoxides or cyclopropanes under mild conditions. In synthetic organic chemistry, trimethylsulfoxonium iodide is valued for its stability and ease of handling compared with other ylide precursors. Its solid crystalline form and high solubility in polar aprotic solvents make it convenient for laboratory use. Upon deprotonation, the resulting sulfoxonium ylide acts as a nucleophile, reacting with aldehydes or ketones to form epoxides stereoselectively. This method has been widely applied in the synthesis of natural products, pharmaceuticals, and complex molecules that contain epoxide or cyclopropane motifs, which serve as key intermediates in further functionalizations. Beyond its role in epoxidation, trimethylsulfoxonium iodide has also been applied in cyclopropanation reactions. By reacting with electron-deficient alkenes, the sulfoxonium ylide can form cyclopropane rings with predictable stereochemistry. These transformations are particularly useful in the preparation of biologically active compounds and fine chemicals, where precise control over stereochemistry is critical. The compound’s reactivity is largely governed by the electron-rich nature of the ylide and the steric effects imparted by the methyl substituents, which influence both reaction rate and selectivity. The preparation of trimethylsulfoxonium iodide typically involves the alkylation of dimethyl sulfoxide (DMSO) with methyl iodide under controlled conditions. This straightforward synthesis allows for efficient production of the reagent on both laboratory and industrial scales. The iodide salt is preferred for many applications due to its solubility properties and the ease of generating the corresponding ylide under basic conditions. Trimethylsulfoxonium iodide continues to be an important reagent in modern organic synthesis due to its versatility, high reactivity, and compatibility with a range of functional groups. Its use in epoxidation and cyclopropanation reactions has enabled the construction of complex molecular architectures that are essential in medicinal chemistry, agrochemicals, and natural product synthesis. The development of this reagent exemplifies the utility of onium salts in facilitating highly selective and efficient chemical transformations. |
| Market Analysis Reports |