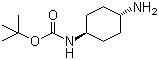
N-Boc-trans-1,4-cyclohexanediamine is an important synthetic intermediate that has been widely used in organic and medicinal chemistry, particularly in the construction of complex molecules and drug candidates. Structurally, it consists of a cyclohexane ring substituted at the 1- and 4-positions with amino groups, where one amino group is protected with a tert-butoxycarbonyl (Boc) group. The trans configuration ensures that the two substituents are on opposite sides of the cyclohexane plane, which significantly affects the molecule's chemical behavior and biological interactions.
The discovery and preparation of N-Boc-trans-1,4-cyclohexanediamine emerged from efforts to develop building blocks that could impart specific three-dimensional shapes to molecules. The trans-1,4-disubstituted cyclohexane scaffold provides a rigid, well-defined spatial arrangement that is advantageous for designing ligands, inhibitors, and other bioactive compounds. Protecting one of the amines with a Boc group allows selective manipulation of the free amine, offering greater control in sequential chemical transformations.
Applications of N-Boc-trans-1,4-cyclohexanediamine are particularly prominent in the synthesis of peptidomimetics, where it serves as a non-natural amino acid surrogate. Its incorporation into peptide backbones can enhance metabolic stability, improve membrane permeability, and introduce conformational constraints that mimic bioactive secondary structures. This approach has been valuable in the development of therapeutic peptides and inhibitors of protein-protein interactions.
In addition to peptidomimetics, N-Boc-trans-1,4-cyclohexanediamine has found use in the preparation of small molecule drugs targeting a variety of biological systems. Its rigid trans-cyclohexane structure can favor specific binding orientations within enzyme active sites or receptor pockets, increasing potency and selectivity. Medicinal chemists have exploited this property in designing kinase inhibitors, protease inhibitors, and modulators of ion channels.
The compound also plays a role in materials science, where it has been utilized as a monomer in the synthesis of polyamides and polyureas. The resulting polymers benefit from the rigidity imparted by the cyclohexane core, leading to materials with enhanced mechanical strength and thermal stability. Such polymers have applications in coatings, adhesives, and biomedical devices.
From a synthetic standpoint, the Boc protecting group in N-Boc-trans-1,4-cyclohexanediamine provides significant versatility. It can be removed under mildly acidic conditions without affecting other sensitive functional groups, enabling sequential and selective transformations. This property makes the molecule particularly suitable for use in solid-phase synthesis strategies, such as those employed in combinatorial chemistry and library generation.
Further functionalization of N-Boc-trans-1,4-cyclohexanediamine often involves alkylation, acylation, or sulfonylation reactions at the free amine, allowing for rapid diversification. The spatial separation of the two amino groups, combined with the ability to protect and deprotect selectively, offers chemists a powerful tool for constructing molecules with precise three-dimensional architectures.
Overall, N-Boc-trans-1,4-cyclohexanediamine exemplifies the type of multifunctional intermediate that has been crucial in advancing both synthetic methodology and applied research in medicinal chemistry. Its discovery and continued use highlight the importance of structural control in the design of bioactive molecules and functional materials.
|



































 GHS09 Details
GHS09 Details