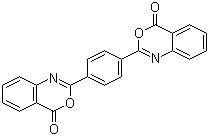
2,2'-Benzene-1,4-diylbis(4H-3,1-benzoxazin-4-one) is an organic compound that features a distinctive structure consisting of a benzene-1,4-diyl (phenyl) group linked to two 3,1-benzoxazin-4-one moieties. This chemical is part of the broader class of benzoxazinones, which have garnered attention for their interesting chemical properties and potential applications in various fields, including materials science, organic electronics, and drug design. The discovery of 2,2'-benzene-1,4-diylbis(4H-3,1-benzoxazin-4-one) represents an advancement in the study of functionalized aromatic compounds and their role in developing novel materials and bioactive molecules.
The synthesis of 2,2'-benzene-1,4-diylbis(4H-3,1-benzoxazin-4-one) typically involves the condensation of appropriate precursor compounds under controlled conditions, ensuring that the desired benzoxazinone subunits are attached to the benzene core. The reaction is carefully designed to provide a high yield while maintaining the stability of the benzoxazinone rings. The resulting compound has both the structural stability of the benzene ring and the reactivity of the benzoxazinone units, making it a valuable building block for the synthesis of more complex materials.
One of the primary applications of 2,2'-benzene-1,4-diylbis(4H-3,1-benzoxazin-4-one) lies in its potential use in organic electronics, particularly in organic semiconductors and light-emitting diodes (OLEDs). The compound's conjugated structure, which includes multiple aromatic rings, allows for efficient charge transport and emission properties, making it suitable for use in optoelectronic devices. Organic semiconductors are an area of significant interest due to their potential to provide flexible, lightweight, and cost-effective alternatives to traditional inorganic semiconductors. The benzoxazinone groups within the structure contribute to the material's ability to absorb and emit light efficiently, which is essential for the development of OLEDs and other optoelectronic applications.
In addition to its role in organic electronics, 2,2'-benzene-1,4-diylbis(4H-3,1-benzoxazin-4-one) has been explored for its potential bioactive properties. Benzoxazinone derivatives are known to exhibit a range of biological activities, including antimicrobial, anticancer, and anti-inflammatory effects. The compound's ability to interact with biological systems makes it a promising candidate for use in the pharmaceutical industry. Research into the pharmacological properties of benzoxazinone derivatives continues to grow, with the aim of developing novel therapeutic agents based on these structures.
Furthermore, the compound has been investigated for use in the design of advanced materials. The unique combination of aromatic and heterocyclic moieties in its structure gives 2,2'-benzene-1,4-diylbis(4H-3,1-benzoxazin-4-one) the potential for use in specialized coatings, catalysts, and other functional materials. The ability of benzoxazinone derivatives to form strong interactions with various substrates can be leveraged to create materials with specific chemical or mechanical properties. For example, these materials could be used in applications where high thermal stability, corrosion resistance, or enhanced chemical reactivity are required.
The discovery and development of 2,2'-benzene-1,4-diylbis(4H-3,1-benzoxazin-4-one) highlight the growing interest in functionalized benzoxazinone compounds. These materials offer exciting possibilities for the development of next-generation organic electronic devices, advanced materials, and novel therapeutic agents.
References
none
|


























 GHS07 Warning Details
GHS07 Warning Details