Online Database of Chemicals from Around the World
| XinChem Corporation | China | |||
|---|---|---|---|---|
 | www.xinchem.com | |||
 | +86 (21) 3412-3252 | |||
 | +86 (21) 5415-3973 | |||
 | xinchem@126.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Haimen RuiYi Medical Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ruiyitech.cn | |||
 | +86 (21) 5471-1706 | |||
 | +86 (21) 5471-1707 | |||
 | sales@ruiyitech.com zyj3625@msn.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Nanjing SiSiB Silicones Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sisib.com | |||
 | +86 (25) 5859-9930 5859-9931 5859-9932 +86 13913891929 | |||
 | +86 (25) 5859-9935 | |||
 | sales@sisib.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Dalchem | Russia | |||
|---|---|---|---|---|
 | www.dalchem.com | |||
 | +7 (8312) 753-772 | |||
 | +7 (8312) 750-799 | |||
 | dregichy@dalchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Classification | Chemical reagent >> Organic reagent >> Silane |
|---|---|
| Name | Tris(trimethylsilyl)silane |
| Molecular Structure | 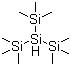 |
| Molecular Formula | C9H27Si4 |
| Molecular Weight | 247.65 |
| CAS Registry Number | 1873-77-4 |
| EC Number | 678-629-0 |
| SMILES | C[Si](C)(C)[SiH]([Si](C)(C)C)[Si](C)(C)C |
| Density | 0.806 |
|---|---|
| Boiling point | 73 °C (5 mmHg) |
| Refractive index | 1.489 |
| Flash point | 55 °C |
| Hazard Symbols |   GHS02;GHS07 Danger Details GHS02;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H315-H319 Details | ||||||||||||||||||||||||
| Safety Statements | P501-P240-P210-P233-P243-P241-P242-P264-P280-P370+P378-P337+P313-P305+P351+P338-P362+P364-P303+P361+P353-P332+P313-P403+P235 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Transport Information | UN 1993 | ||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Tris(trimethylsilyl)silane, commonly abbreviated as TTMS or (TMS)₃SiH, is an organosilicon compound that has become an important reagent in organic synthesis due to its unique properties as a free radical mediator and hydrogen donor. Tris(trimethylsilyl)silane was first reported in the early 1980s by researchers seeking a safer and more efficient hydrogen donor than traditional reagents such as tin hydrides. Its discovery provided chemists with a new tool, especially in free radical chemistry where control and selectivity of reduction processes are crucial. Its chemical structure is a central silicon atom bonded to three trimethylsilyl (TMS) groups and a hydrogen atom, and the Si-H bonds in TTMS act as a source of hydrogen atoms in a variety of chemical reactions. It is a colorless liquid that is soluble in common organic solvents such as toluene, hexane, and THF. TTMS is widely used as a free radical hydrogen donor in organic synthesis. Its ability to efficiently transfer hydrogen atoms makes it well suited for free radical reduction reactions, including the reduction of alkyl and aryl halides, the dehalogenation of polyhalogenated compounds, and the reduction of alkynes to alkenes. The use of TTMS allows for selective reductions under mild conditions, avoiding the harsh reagents and conditions typically required by traditional methods. This selectivity is particularly valuable in complex molecular synthesis, where it is critical to maintain the integrity of functional groups. In the desulfurization of thioketones and other sulfur-containing compounds, TTMS serves as a clean and efficient hydrogen source to convert these substrates to the corresponding hydrocarbons without generating toxic byproducts. TTMS is used for the deprotection of sensitive functional groups, such as the cleavage of silyl ethers, without affecting other functional groups in the molecule. This property is highly advantageous in multi-step synthetic routes where protecting groups are strategically used. In complex molecular synthesis, TTMS is used to promote free radical-mediated transformations, such as carbon-carbon and carbon-heteroatom bond formation. Its use improves reaction efficiency and yields, especially in the synthesis of natural products and pharmaceuticals. TTMS is used as a free radical initiator in polymer chemistry to aid in the polymerization of monomers and the modification of polymer structures, aiding the development of advanced materials with tailored properties. TTMS offers a more environmentally friendly alternative to traditional reagents, such as tin hydrides, which are toxic and generate hazardous waste. Its application reduces the environmental impact of chemical processes and improves the safety of synthetic operations. In a research setting, TTMS is used to study free radical mechanisms, gaining insights into free radical behavior and the development of new free radical-based reactions. This knowledge has advanced the field of free radical chemistry and broadened the range of synthetic methods. Ongoing research explores new applications of TTMS in a variety of chemical processes, including its role in catalysis, materials science, and emerging technologies. References 2024. Boryl radical-mediated halogen-atom transfer enables arylation of alkyl halides with electrophilic and nucleophilic coupling partners. Nature Synthesis. DOI: 10.1038/s44160-024-00587-5 2024. Total synthesis of Comfreyn A and structural analogues via two photochemical key steps. Photochemical & photobiological sciences : Official journal of the European Photochemistry Association and the European Society for Photobiology. DOI: https://pubmed.ncbi.nlm.nih.gov/38935211 2023. Catalytic acceptorless dehydrogenative coupling mediated by photoinduced hydrogen-atom transfer. Nature Synthesis. DOI: 10.1038/s44160-022-00195-1 |
| Market Analysis Reports |