Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| 2A Pharmachem USA | USA | |||
|---|---|---|---|---|
 | www.2apharmachem.com | |||
 | +1 (630) 322-8887 | |||
 | +1 (630) 322-8885 | |||
 | sales@2abiotech.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Hohance Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hohance.com | |||
 | +86 (21) 3111-5312 | |||
 | +86 (21) 3111-5317 | |||
 | info@hohance.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hubei Tuoyuan Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemty.com | |||
 | +86 (027) 8470-0466 | |||
 | +86 (027) 8470-0466 | |||
 | 59741184@qq.com | |||
 | QQ Chat | |||
 | WeChat: 13260668040 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Apexbio Technology LLC | USA | |||
|---|---|---|---|---|
 | www.apexbt.com | |||
 | +1 (832) 696-8203 | |||
 | +1 (855) 527-3928 | |||
 | info@apexbt.com | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Selleck Chemicals LLC | USA | |||
|---|---|---|---|---|
 | www.selleckchem.com | |||
 | +1 (713) 535-9129 | |||
 | +1 (832) 582-8590 | |||
 | info@selleckchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Cfm Oskar Tropitzsch GmbH | Germany | |||
|---|---|---|---|---|
 | www.cfmot.de | |||
 | +49 (9231) 9619-0 | |||
 | +49 (9231) 9619-60 | |||
 | info@cfmot.de | |||
| Chemical distributor since 1985 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Generay Biotech (Shanghai) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.generay.com.cn | |||
 | +86 (21) 6762-6003 6762-6050 6762-6120 | |||
 | +86 (21) 6762-6011 | |||
 | order@generay.com.cn | |||
| Chemical manufacturer | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Shanghai Fine Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sfcc-chem.com | |||
 | +86 (21) 3358-1117 3358-1118 3358-1119 | |||
 | +86 (21) 3358-1128 | |||
 | sfcc@sfcc-chem.com | |||
| Chemical manufacturer | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antibiotics >> Other antibiotics |
|---|---|
| Name | Streptozocin |
| Synonyms | 2-Desoxy-2-(3-methyl-3-nitrosoureido)-D-glucopyranose; 2-Deoxy-2-[[(methylnitrosoamino)carbonyl]amino]-D-glucose |
| Molecular Structure | 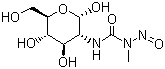 |
| Molecular Formula | C8H15N3O7 |
| Molecular Weight | 265.22 |
| CAS Registry Number | 18883-66-4 |
| EC Number | 242-646-8 |
| SMILES | CN(C(=O)N[C@@H]1[C@H]([C@@H]([C@H](O[C@@H]1O)CO)O)O)N=O |
| Density | 1.9±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 121 °C (Decomposes) (Expl.) |
| Solubility | 10 mM in DMSO (Expl.) |
| Index of refraction | 1.67 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H312-H332-H340-H350-H360 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P271-P280-P302+P352-P304+P340-P317-P318-P321-P362+P364-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Streptozocin is a naturally occurring nitrosourea compound isolated from the bacterium Streptomyces achromogenes. It was first discovered in the 1950s during the search for novel antitumor antibiotics. The compound gained attention for its unique chemical structure, which combines a glucose moiety with a nitrosourea functional group, enabling it to target specific cells selectively. Streptozocin has been primarily developed and applied as a chemotherapeutic agent. Its mechanism of action involves alkylation and DNA strand breakage, leading to cytotoxic effects, particularly in cells with high glucose uptake. This selectivity allows streptozocin to preferentially target pancreatic beta cells, which absorb glucose at a high rate. Consequently, streptozocin has been widely used in oncology for the treatment of pancreatic islet cell tumors, also known as pancreatic neuroendocrine tumors. Beyond its clinical application in oncology, streptozocin has been employed in experimental research as a diabetogenic agent. Due to its selective toxicity to pancreatic beta cells, administration of streptozocin in laboratory animals induces insulin-dependent diabetes mellitus. This model is extensively utilized in studies of diabetes pathophysiology and in the evaluation of antidiabetic drugs. Clinically, streptozocin is often administered in combination with other chemotherapeutic agents to enhance antitumor efficacy. It is given intravenously, and its pharmacological profile includes rapid uptake into target cells, metabolic activation, and generation of DNA-damaging species. Its use is accompanied by monitoring for side effects such as nephrotoxicity, hepatotoxicity, and nausea. Streptozocin’s discovery provided a valuable tool both for cancer therapy and for diabetes research, contributing to advances in understanding pancreatic function and disease. Its dual role highlights the importance of natural products in drug discovery and biomedical research. The chemical’s stability and solubility characteristics have allowed for various formulation developments, improving its clinical usability. Ongoing research explores analogues and derivatives of streptozocin aiming to optimize therapeutic indices and reduce adverse effects. Overall, streptozocin remains an important compound in both clinical and research settings due to its unique biological activity and specificity for pancreatic beta cells. References 1994. Comparison of the effects of various vanadium salts on glucose homeostasis in streptozotocin-diabetic rats. European Journal of Pharmacology, 261(1-2). DOI: 10.1016/0014-2999(94)90334-4 1998. Streptozotocin may provide protection against subsequent oxidative stress of endotoxin or streptozotocin in rats. Journal of Biochemical and Molecular Toxicology, 12(3). DOI: 10.1002/(sici)1099-0461(1998)12:3<143::aid-jbt2>3.0.co;2-l 2024. Antihyperglycemic and hypolipidemic effect of yellow-fleshed cassava/wheat flour composite bread in high fat/streptozotocin induced diabetic rats. Journal of Food Measurement and Characterization. DOI: 10.1007/s11694-024-02622-7 |
| Market Analysis Reports |