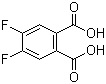
4,5-Difluorophthalic acid is an organic compound belonging to the class of fluorinated aromatic acids. It consists of a phthalic acid structure, which is a benzene ring with two carboxylic acid groups (-COOH) attached at the 1,2-positions. In 4,5-difluorophthalic acid, two fluorine atoms are substituted at the 4 and 5 positions on the aromatic ring, which significantly alters its chemical and physical properties.
The discovery and synthesis of 4,5-difluorophthalic acid have been part of a broader effort to study the effects of fluorination on the properties of organic compounds. The introduction of fluorine atoms into aromatic systems is known to influence a wide range of chemical characteristics, including increased stability, altered solubility, and changes in reactivity. This makes fluorinated compounds like 4,5-difluorophthalic acid of particular interest in various fields, including materials science, medicinal chemistry, and polymer chemistry.
4,5-Difluorophthalic acid is typically synthesized through fluorination reactions, where fluorine atoms are introduced into the phthalic acid structure. The introduction of fluorine can be accomplished through electrophilic aromatic substitution reactions, a common method for introducing fluorine onto an aromatic ring. The specific placement of the fluorine atoms at the 4 and 5 positions of the aromatic ring is important for the compound's electronic properties, which may influence its reactivity and interaction with other chemical species.
In terms of applications, 4,5-difluorophthalic acid has been explored as a precursor to various fluorinated materials. Its unique fluorine-substituted structure can impart desirable properties to materials, such as improved chemical stability, higher resistance to thermal degradation, and better resistance to environmental factors. These attributes make 4,5-difluorophthalic acid and its derivatives suitable for use in coatings, polymers, and other materials where durability and stability are required.
Additionally, the fluorination of aromatic acids like phthalic acid is also relevant to the development of high-performance materials in the electronics industry. Fluorinated compounds are often used in the production of polymers for electronic applications, such as in the manufacturing of semiconductors, insulators, and other electronic components. The use of 4,5-difluorophthalic acid in the synthesis of such materials can help enhance their performance by improving their chemical and thermal stability.
In medicinal chemistry, fluorinated aromatic compounds are of particular interest due to the influence of fluorine on the biological properties of drugs. The addition of fluorine atoms to a molecule can modify its lipophilicity, metabolic stability, and ability to bind to biological targets. While 4,5-difluorophthalic acid itself may not be directly used as a drug, its derivatives or related compounds could potentially be explored for their biological activities. Fluorinated aromatic acids are often investigated for their antibacterial, antiviral, and anticancer properties, as the fluorine atoms can alter the interactions between the compound and biological molecules.
Moreover, 4,5-difluorophthalic acid can serve as a building block for the synthesis of other fluorinated organic compounds, which can be used in a variety of industrial and research applications. By modifying the carboxyl groups or the fluorine atoms, it is possible to create a wide range of derivatives with tailored properties for specific applications.
In conclusion, 4,5-difluorophthalic acid is a valuable compound in the field of fluorinated organic chemistry. Its unique fluorine-substituted structure provides a range of applications in materials science, electronics, and potentially in medicinal chemistry. The ability to incorporate fluorine into aromatic acid structures opens up possibilities for the development of high-performance materials and bioactive compounds with enhanced stability and specificity.
|









































 GHS07 Warning Details
GHS07 Warning Details