Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Organic sulfur compound |
|---|---|
| Name | Lawesson's Reagent |
| Synonyms | 2,4-Bis(4-methoxyphenyl)-1,3-dithia-2,4-diphosphetane-2,4-disulfide |
| Molecular Structure | 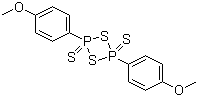 |
| Molecular Formula | C14H14O2P2S4 |
| Molecular Weight | 404.47 |
| CAS Registry Number | 19172-47-5 |
| EC Number | 242-855-4 |
| SMILES | COC1=CC=C(C=C1)P2(=S)SP(=S)(S2)C3=CC=C(C=C3)OC |
| Melting point | 228-231 °C |
|---|---|
| Water solubility | decomposes |
| Hazard Symbols |   GHS02;GHS07 Danger Details GHS02;GHS07 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H261-H302-H312-H332 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P231+P232-P261-P264-P270-P271-P280-P301+P317-P302+P352-P304+P340-P317-P321-P330-P362+P364-P370+P378-P402+P404-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Lawesson's reagent is a well-known sulfurizing agent widely used in organic synthesis to convert carbonyl compounds into their thio analogs. Lawesson's reagent has the chemical formula [(CH₃C₆H₄)₂P₂S₄] and was discovered by Swedish chemist Sven-Olov Lawesson in 1956 while exploring phosphorus-sulfur chemistry. Lawesson's search for an efficient method to synthesize thioesters and other sulfur-containing compounds led to the identification of the reagent. Its structure consists of a phosphorus-sulfur core and two p-methylphenyl groups. The two phosphorus atoms are connected by a sulfur atom to form a P₂S₄ ring, and two aromatic rings are attached to the phosphorus for enhanced stability. It is a yellow crystalline solid that is soluble in organic solvents such as dichloromethane and toluene. Lawesson's reagent is mainly used to convert carbonyl compounds such as ketones and aldehydes into the corresponding thiocarbonyl derivatives such as thiones and thioesters. This transformation is essential for the synthesis of sulfur-containing heterocycles and biologically active molecules. It is also effective in converting amides to thioamides, which are valuable intermediates in the synthesis of drugs and natural products. Lawesson's reagent facilitates the synthesis of thiazoles and thiophenes by inserting sulfur into carbon-nitrogen bonds and carbon-carbon bonds, respectively. These heterocycles are important scaffolds in medicinal chemistry and materials science. It is also used to construct thiopyran and thiorolidine rings, which are components of various natural products and synthetic drugs. In medicinal chemistry, Lawesson's reagent is used to synthesize sulfur analogs of drugs. This modification can enhance the biological activity, stability, and pharmacokinetic properties of the parent compound. It is used to make thiopeptides and other sulfur-containing peptide analogs as protease inhibitors. These compounds are essential for the development of therapeutics for diseases associated with protease activity. Lawesson's reagent facilitates the synthesis of sulfur-containing polymers with unique electrical properties. These polymers are used in electronic devices, sensors, and batteries. The reagent is used to produce thio- and dithio-dyes, which have applications in the textile and photonic industries due to their stability and color properties. Lawesson's reagent is used to introduce thioesters and other protecting groups in organic synthesis. These groups can be selectively removed or transformed in subsequent steps, allowing controlled chemical reactions. It allows the interconversion of functional groups such as ketones, amides, and esters into their thio-analogs, providing chemists with tools to modify molecular structures. References 2021. Lawesson’s Reagent. Name Reactions, 83. DOI: 10.1007/978-3-030-50865-4_83 2021. Thionation of Aminophthalimide Hindered Carbonyl Groups and Application to the Synthesis of 3,6’-Dithionated Pomalidomides. Synlett, 32(8). DOI: 10.1055/s-0040-1720460 2021. S-Heterocycle Synthesis. Lawesson’s Reagent in Heterocycle Synthesis, 6. DOI: 10.1007/978-981-16-4655-3_6 |
| Market Analysis Reports |