Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Classification | Chemical reagent >> Organic reagent >> Hydrazine |
|---|---|
| Name | 4-(Dimethylamino)benzoylhydrazide |
| Synonyms | 4-(Dimethylamino)benzocarbohydrazide; 4-(Dimethylamino)benzohydrazide; 4-(Dimethylamino)benzoylhydrazine |
| Molecular Structure | 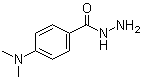 |
| Molecular Formula | C9H13N3O |
| Molecular Weight | 179.22 |
| Protein Sequence | X |
| CAS Registry Number | 19353-92-5 |
| EC Number | 242-985-1 |
| SMILES | CN(C)C1=CC=C(C=C1)C(=O)NN |
| Density | 1.2±0.1 g/cm3, Calc.*, 1.156 g/mL |
|---|---|
| Melting point | 174-175 °C |
| Index of Refraction | 1.601, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H317-H319-H335-H351 Details | ||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P264-P264+P265-P270-P271-P272-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P318-P319-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
4-(Dimethylamino)benzoylhydrazide, a synthetic organic compound, has garnered attention for its diverse applications, particularly in the field of organic chemistry and pharmaceuticals. Its discovery can be attributed to ongoing research in the mid-20th century, where scientists were exploring various hydrazine derivatives for their potential biological activities. The compound features a hydrazide functional group attached to a dimethylaminobenzoyl moiety, which contributes to its unique reactivity and properties. The synthesis of 4-(dimethylamino)benzoylhydrazide typically involves the reaction of 4-(dimethylamino)benzoic acid with hydrazine or its derivatives, resulting in the formation of the hydrazide through a condensation reaction. This compound is characterized by its ability to participate in various chemical reactions, including acylation and condensation reactions, making it a versatile intermediate in organic synthesis. One of the primary applications of 4-(dimethylamino)benzoylhydrazide lies in its use as a reagent in organic synthesis. Its reactive hydrazide functional group allows it to form hydrazones and azo compounds, which are important intermediates in the synthesis of dyes, pigments, and pharmaceuticals. The ability to generate a wide range of derivatives makes this compound a valuable building block for creating more complex molecular structures. In the pharmaceutical industry, 4-(dimethylamino)benzoylhydrazide has been investigated for its potential biological activities. Studies have indicated that compounds derived from this hydrazide exhibit antimicrobial, antifungal, and anti-inflammatory properties, suggesting that they may serve as lead compounds in drug development. Researchers have focused on modifying its structure to enhance its biological efficacy and reduce toxicity, exploring its potential in treating various diseases. Moreover, the compound has found applications in analytical chemistry as a reagent for the detection and quantification of certain metal ions. The presence of the dimethylamino group enhances its reactivity, allowing it to form colored complexes with metal ions, facilitating their determination in different samples. This application is particularly useful in environmental analysis and quality control processes in various industries. Despite its promising applications, the use of 4-(dimethylamino)benzoylhydrazide requires careful consideration of its safety and environmental impact. Researchers are investigating its toxicity and potential ecological effects to ensure that its use aligns with regulatory guidelines and environmental sustainability. In summary, 4-(dimethylamino)benzoylhydrazide is a significant chemical compound with diverse applications in organic synthesis, pharmaceuticals, and analytical chemistry. Its discovery marked a notable advancement in the development of hydrazine derivatives, leading to various important applications. As research continues, the compound holds promise for further exploration in drug development and industrial processes. References 2023. Screening a small hydrazide-hydrazone combinatorial library for targeting the STAT3 in monocyte-macrophages with insulated reporter transposons. Medicinal Chemistry Research, 32(3). DOI: 10.1007/s00044-023-03028-8 2022. Discovery of small molecule inhibitors of Plasmodium falciparum apicoplast DNA polymerase. Journal of Enzyme Inhibition and Medicinal Chemistry, 37(1). DOI: 10.1080/14756366.2022.2070909 2020. Synthesis and Spectral-Luminescent Properties of 2-Aryl-5-butyl-1,3,4-oxadiazoles. Russian Journal of General Chemistry, 90(11). DOI: 10.1134/s1070363220110079 |
| Market Analysis Reports |