Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Henan Ouber Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oubertec.com | |||
 | +86 (371) 6532-2607 +86 18937141980 | |||
 | +86 (371) 6532-2607 | |||
 | anna.zhang@oubertec.com | |||
 | QQ Chat | |||
 | WeChat: 18937141980 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Isoquinoline compound |
|---|---|
| Name | 1-(3,5-Dimethylphenyl)-6-(1-methylethyl)isoquinoline |
| Molecular Structure | 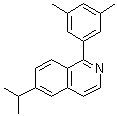 |
| Molecular Formula | C20H21N |
| Molecular Weight | 275.39 |
| CAS Registry Number | 1936437-58-9 |
| SMILES | CC1=CC(=CC(=C1)C2=NC=CC3=C2C=CC(=C3)C(C)C)C |
| Density | 1.0±0.1 g/cm3, Calc.*Solubility |
|---|---|
| Density | 1.040±0.06 g/cm3 (20 °C 760 Torr), Calc.*1.601, Calc.* |
| Boiling Point | 394.6±11.0 °C (760 mmHg), Calc.* |
| Flash Point | 168.5±12.0 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2020 ACD/Labs) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
|
1-(3,5-Dimethylphenyl)-6-(1-methylethyl)isoquinoline is a compound belonging to the class of isoquinoline derivatives. It is a member of a larger group of organic molecules that possess various aromatic and heterocyclic structures. This particular compound contains both a substituted phenyl group (with methyl groups at positions 3 and 5) and an isopropyl group at the 6-position of the isoquinoline ring. The discovery of isoquinoline derivatives, including 1-(3,5-dimethylphenyl)-6-(1-methylethyl)isoquinoline, stems from ongoing research into the structural diversity and reactivity of nitrogen-containing heterocycles. Isoquinolines have been recognized for their role in medicinal chemistry, primarily due to the biological activities exhibited by compounds in this family. Over the years, numerous isoquinoline derivatives have been studied for their potential therapeutic properties, which has significantly contributed to the development of this chemical class. The synthesis of 1-(3,5-dimethylphenyl)-6-(1-methylethyl)isoquinoline is typically achieved through standard synthetic organic chemistry techniques, such as Friedlander condensation or other cyclization reactions that involve the coupling of appropriate aromatic precursors. The presence of both methyl and isopropyl groups in the structure suggests that this compound is likely designed for specific interactions, either through steric effects or through its electronic properties, which may influence its reactivity or interactions with biological targets. 1-(3,5-Dimethylphenyl)-6-(1-methylethyl)isoquinoline, as a representative of isoquinoline derivatives, finds its most significant applications in medicinal and pharmaceutical chemistry. Isoquinoline derivatives are known to exhibit a wide range of biological activities, including antimicrobial, anticancer, anti-inflammatory, and analgesic effects. Due to their structural similarity to naturally occurring alkaloids, many isoquinoline derivatives are explored for their potential as drug candidates. This compound could, therefore, be a candidate for similar applications, potentially contributing to the design of new pharmaceutical agents. In addition to its medicinal applications, 1-(3,5-dimethylphenyl)-6-(1-methylethyl)isoquinoline may also be investigated for use in other chemical fields, such as material science or as a ligand in metal-catalyzed reactions. Its aromatic and heterocyclic structure could potentially contribute to the development of specialized materials or catalysts with particular electronic or physical properties. Overall, 1-(3,5-dimethylphenyl)-6-(1-methylethyl)isoquinoline represents an interesting class of isoquinoline derivatives, which have been the subject of research due to their broad potential in medicinal chemistry and other fields of applied science. However, its exact role and applications in various chemical processes continue to be studied within the broader context of isoquinoline chemistry. References none |
| Market Analysis Reports |