Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shanghai Amsheng Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amsheng.com | |||
 | +86 (21) 6267-3642 | |||
 | +86 (21) 6218-0706 | |||
 | info@amsheng.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Manus Aktteva Biopharma Llp | China | |||
|---|---|---|---|---|
 | www.manusaktteva.com | |||
 | +91 ((79) 2646-3395 | |||
 | +91 ((79) 2646-3395 | |||
 | staff@manusakttevabiopharma.in | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Xiamen Kaijia Imp & Exp Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kaijiachem.com | |||
 | +86 (592) 510-1177 | |||
 | +86 (592) 565-3685 | |||
 | jojo@kaijiachem.com kaijiachem@163.com | |||
| Chemical distributor since 2006 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| La Mesta Chimie Fine S.A.S. | France | |||
|---|---|---|---|---|
 | www.la-mesta.com | |||
 | +33 (4) 9208-5300 | |||
 | +33 (4) 9208-5340 | |||
 | info@la-mesta.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Srigen Lifesciences Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.srigenlifesciences.com | |||
 | +91 (40) 2307-1899 | |||
 | +91 (40) 2307-1899 | |||
 | info@srigenlifesciences.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Ce Pharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cepharm.cn | |||
 | +86 (21) 2097-0199 | |||
 | binbin.lin@cepharm.cn | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Saflik Pharma LLP | India | |||
|---|---|---|---|---|
 | www.saflikpharma.com | |||
 | +91 7600062602 | |||
 | info@saflikpharma.com | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Kingchem Inc. | USA | |||
|---|---|---|---|---|
 | www.kingchem.com | |||
 | +1 (800) 211-4330 (201) 825-9988 | |||
 | +1 (201) 825-9148 | |||
 | customer-service@kingchem.com | |||
| Chemical manufacturer since 1994 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Jiangxi Fangzun Pharmaceutical & Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fangzunchem.com | |||
 | +86 15961115886 | |||
 | sales@baokangchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| Cambrex Corporation | USA | |||
|---|---|---|---|---|
 | www.cambrex.com | |||
 | +1 (201) 804-3000 | |||
 | +1 (201) 804-9852 | |||
 | info@cambrex.com | |||
| Chemical manufacturer | ||||
| True Pharmachem Inc. | USA | |||
|---|---|---|---|---|
 | www.truepharmachem.com | |||
 | +1 (217) 766-6932 | |||
 | +1 (973) 588-3470 | |||
 | sales@truepharmachem.com | |||
| Chemical manufacturer | ||||
| Zhejiang Tianxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.txpharm.com | |||
 | +86 (576) 8399-3179 8399-3699 8399-3659 | |||
 | +86 (576) 8399-3869 | |||
 | sales@txpharm.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Hydrocarbon nitrite |
|---|---|
| Name | 2-Methyl-3-nitrobenzoic acid |
| Synonyms | 3-Nitro-o-toluic acid |
| Molecular Structure | 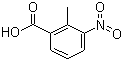 |
| Molecular Formula | C8H7NO4 |
| Molecular Weight | 181.15 |
| CAS Registry Number | 1975-50-4 |
| EC Number | 217-826-4 |
| SMILES | CC1=C(C=CC=C1[N+](=O)[O-])C(=O)O |
| Density | 1.4±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 182-184 °C (Expl.) |
| Index of Refraction | 1.601, Calc.* |
| Boiling Point | 337.1±30.0 °C (760 mmHg), Calc.* |
| Flash Point | 151.9±13.0 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
2-Methyl-3-nitrobenzoic acid is an organic compound that belongs to the class of substituted benzoic acids. It consists of a benzene ring with two substituents: a methyl group at the second position and a nitro group at the third position. The compound is also known by other names such as 3-nitro-o-toluic acid. The discovery of 2-methyl-3-nitrobenzoic acid follows the historical development of substituted benzoic acids, a group of compounds that has been extensively studied due to their widespread presence in organic chemistry and their diverse reactivity. This compound is one of many examples of substituted aromatic acids, which have long been of interest in both industrial and academic chemistry due to their versatile reactivity and applications. The synthesis of 2-methyl-3-nitrobenzoic acid is typically achieved through nitration of 2-methylbenzoic acid, a process that involves the introduction of a nitro group (-NO2) to the aromatic ring. The nitration reaction typically uses a mixture of concentrated nitric acid and sulfuric acid, which is a standard method for the introduction of nitro groups into aromatic compounds. This synthetic pathway allows for the preparation of the compound in a controlled manner, and such reactions are widely used in the synthesis of various nitrated aromatic compounds. 2-Methyl-3-nitrobenzoic acid has been utilized in several fields of chemistry, particularly in the synthesis of more complex organic molecules. Its reactivity and the presence of both a carboxyl group (-COOH) and a nitro group (-NO2) make it useful as an intermediate in the synthesis of other chemicals, including dyes, pharmaceuticals, and agrochemicals. It may also be involved in the preparation of other substituted benzoic acids or aromatic compounds with more elaborate functional groups. The compound has potential applications in the synthesis of herbicides and other agrochemicals. Many nitro-substituted aromatic acids and their derivatives are known for their biological activity, which can influence various agricultural and environmental processes. Additionally, it can serve as a precursor for the synthesis of certain drugs and compounds used in chemical research. While 2-methyl-3-nitrobenzoic acid has been explored for various uses, its primary significance remains in the context of synthetic organic chemistry, where it serves as an important building block for the creation of more complex molecules. The presence of the nitro group allows for further chemical modifications, enabling the development of a range of chemical products and facilitating the study of reaction mechanisms involving aromatic compounds. Overall, 2-methyl-3-nitrobenzoic acid is a well-established compound in the field of organic chemistry, valued for its role as an intermediate in the synthesis of other chemical substances and its utility in various applications, particularly in the creation of functionalized aromatic compounds. References 1998. Synthesis of antineoplaston A10 analogs as potential antitumor agents. Archives of Pharmacal Research, 21(2). DOI: 10.1007/bf02974021 2002. Determination of 2-methyl-3-nitrobenzoic acid through pretreatment with diazomethane by gas chromatography. Se pu = Chinese Journal of Chromatography, 20(5). PMID: 16358702 2010. (Methanol-κO)(2-methyl-3-nitrobenzoato-κO)triphenyltin(IV). Acta Crystallographica Section E, 66(Pt 9). DOI: 10.1107/s1600536810033623 |
| Market Analysis Reports |