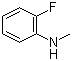
2-Fluoro-N-methylaniline is an organic compound that belongs to the class of anilines, which are aromatic amines characterized by the presence of an amino group attached to a benzene ring. This particular compound features a fluorine atom at the 2-position of the aniline ring and a methyl group attached to the nitrogen atom of the amino group. The discovery of 2-fluoro-N-methylaniline can be traced back to advancements in synthetic organic chemistry, particularly during the mid-20th century when researchers began to explore the potential of fluorinated compounds in various applications. The introduction of fluorine into organic molecules has been shown to alter their chemical properties significantly, enhancing their stability and reactivity.
The synthesis of 2-fluoro-N-methylaniline typically involves the nucleophilic substitution of a suitable precursor, often using fluorinating agents such as hydrogen fluoride or fluorine gas under controlled conditions. This process allows for the selective introduction of the fluorine atom onto the aniline structure, resulting in the desired compound. Various synthetic routes have been developed over the years to improve yield and selectivity, reflecting the growing interest in fluorinated compounds within the scientific community.
2-Fluoro-N-methylaniline has garnered attention in multiple fields, including pharmaceuticals, agrochemicals, and materials science. One of its primary applications is as an intermediate in the synthesis of pharmaceuticals. The introduction of fluorine into drug candidates often enhances their biological activity, metabolic stability, and lipophilicity, leading to improved pharmacokinetic properties. Research has shown that 2-fluoro-N-methylaniline and its derivatives exhibit potential as building blocks in the development of novel therapeutic agents, particularly in treating various diseases such as cancer and bacterial infections.
In the agrochemical industry, 2-fluoro-N-methylaniline is utilized as a precursor in the synthesis of herbicides and pesticides. The incorporation of fluorine into these compounds can enhance their effectiveness, allowing for lower application rates and reduced environmental impact. By modifying the chemical structure of active ingredients, researchers can improve the selectivity and potency of agrochemicals, contributing to more sustainable agricultural practices.
Additionally, 2-fluoro-N-methylaniline finds applications in the development of advanced materials, particularly in the field of polymer chemistry. Fluorinated compounds often exhibit unique properties, such as increased chemical resistance and reduced surface energy. As a result, they can be incorporated into polymer formulations to improve their performance in various applications, including coatings, adhesives, and sealants. The presence of the fluorine atom in 2-fluoro-N-methylaniline can enhance the thermal stability and durability of materials, making them suitable for demanding environments.
Research continues to explore the full potential of 2-fluoro-N-methylaniline and its derivatives in various applications. Scientists are investigating new synthetic methods to create derivatives with tailored properties, enabling the development of specialized compounds for specific uses. The ongoing interest in fluorinated organic compounds highlights their significance in modern chemistry and their potential to address contemporary challenges in pharmaceuticals, agriculture, and materials science.
In summary, 2-fluoro-N-methylaniline is a fluorinated aniline compound with a rich history of discovery and diverse applications in pharmaceuticals, agrochemicals, and materials science. Its unique properties and reactivity make it a valuable building block for the synthesis of a wide range of functional compounds, contributing to advancements in various fields. As research progresses, the potential applications of 2-fluoro-N-methylaniline are expected to expand further, solidifying its importance in contemporary organic chemistry.
References
2017. Synthesis of trisubstituted furans via Ag-catalyzed cascade heterocyclization of 2-(1-alkynyl)-2-alken-1-ones with aryl-amines. Chemical Papers, 71(6). DOI: 10.1007/s11696-017-0323-4
2017. The metabolic pathway of metamifop degradation by consortium ME-1 and its bacterial community structure. Biodegradation, 28(2). DOI: 10.1007/s10532-017-9787-8
|































 GHS07 Warning Details
GHS07 Warning Details