Online Database of Chemicals from Around the World
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Classification | Organic raw materials >> Organic fluorine compound >> Fluorotoluene series |
|---|---|
| Name | 5-Bromo-2-chloro-4-fluorotoluene |
| Synonyms | 1-Bromo-4-chloro-2-fluoro-5-methylbenzene |
| Molecular Structure | 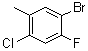 |
| Molecular Formula | C7H5BrClF |
| Molecular Weight | 223.47 |
| CAS Registry Number | 200190-87-0 |
| EC Number | 803-237-5 |
| SMILES | CC1=CC(=C(C=C1Cl)F)Br |
| Solubility | Practically insoluble (0.02 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.618±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 212.0±35.0 °C (760 Torr), Calc.* |
| Flash point | 82.0±25.9 °C, Calc.* |
| Index of refraction | 1.545 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
5-Bromo-2-chloro-4-fluorotoluene is an organic compound belonging to the family of halogenated toluenes. It is characterized by the presence of three halogen atoms—bromine (Br), chlorine (Cl), and fluorine (F)—attached to a toluene backbone, which consists of a methyl group (-CH₃) attached to a benzene ring. Specifically, the structure consists of a bromine atom at the 5-position, a chlorine atom at the 2-position, and a fluorine atom at the 4-position of the benzene ring, making it a highly substituted aromatic compound. The discovery and synthesis of 5-bromo-2-chloro-4-fluorotoluene are part of ongoing efforts in the chemical community to explore the effects of halogen substitution on aromatic compounds. Halogen atoms are often introduced into organic molecules to modify their reactivity, stability, and solubility. The unique combination of bromine, chlorine, and fluorine on the toluene ring imparts distinct properties to this compound, including increased chemical stability and altered electronic characteristics, which can be beneficial for a wide range of applications. This compound can be synthesized via electrophilic aromatic substitution reactions, a common method for halogenating aromatic compounds. In this case, selective halogenation reactions are employed to introduce the desired halogens into specific positions on the benzene ring. The positioning of the halogens influences the compound's electronic distribution, and the presence of different halogens on the ring creates a molecule with distinct reactivity compared to simpler toluenes. 5-Bromo-2-chloro-4-fluorotoluene has found applications in various areas of chemistry and materials science. One key application is in the synthesis of specialty chemicals and intermediates for the production of pharmaceuticals, agrochemicals, and other fine chemicals. The halogenated toluene structure is particularly useful in the synthesis of compounds that require specific electronic properties or functional group reactivity. For example, fluorine substitution can affect the lipophilicity and metabolic stability of compounds, while bromine and chlorine atoms can be used for further chemical transformations. In materials science, 5-bromo-2-chloro-4-fluorotoluene and its derivatives may be utilized as precursors in the development of novel materials with enhanced properties. The halogen substitutions can improve the material's resistance to degradation, increase its stability at high temperatures, or impart unique optical or electrical properties. These characteristics are useful in the design of polymers, coatings, and films, as well as in electronics, where halogenated compounds are often employed to enhance performance or durability. In addition to materials science, the compound can also serve as an important building block in the synthesis of more complex organic molecules. Halogenated toluenes like 5-bromo-2-chloro-4-fluorotoluene are frequently used in the preparation of cross-coupling reagents and catalysts, as well as in the synthesis of heterocyclic compounds and other functionalized organic molecules. Pharmaceutical applications of halogenated toluenes, including 5-bromo-2-chloro-4-fluorotoluene, are another area of interest. Fluorinated compounds, in particular, are widely studied for their impact on the pharmacokinetics and bioactivity of drug molecules. The introduction of halogens such as fluorine can significantly enhance the drug's bioavailability, stability, and ability to interact with biological targets, making such compounds valuable in medicinal chemistry. Although 5-bromo-2-chloro-4-fluorotoluene itself may not have direct therapeutic uses, its derivatives could potentially exhibit pharmacological activity due to the influence of the halogen atoms on their chemical and biological properties. In summary, 5-bromo-2-chloro-4-fluorotoluene is an important halogenated aromatic compound with a range of applications in materials science, pharmaceutical research, and organic synthesis. The presence of multiple halogens on the toluene ring confers unique properties that make this compound valuable for the development of new materials and as a precursor for the synthesis of more complex organic molecules. |
| Market Analysis Reports |