Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Sinfachem Limited | China | |||
|---|---|---|---|---|
 | www.sinfachem.com | |||
 | +86 (25) 8468-3399 8517-8591 | |||
 | +86 (25) 8468-3112 | |||
 | sinfachem@foxmail.com sinfachemltd@foxmail.com info@sinfachem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| SynQuest Labs, Inc. | USA | |||
|---|---|---|---|---|
 | www.synquestlabs.com | |||
 | +1 (386) 462-0788 | |||
 | +1 (386) 462-7097 | |||
 | Sales@synquestlabs.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Aromsyn Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aromsyn.com | |||
 | +86 (571) 8558-5865 +86 13336024896 | |||
 | +86 (571) 8558-5865 | |||
 | service@aromsyn.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Xuchang Chenhe Bio-pharmaceutical Tech. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.the-chem.com | |||
 | +86 (374) 579-3128 +86 15836530076 | |||
 | +86 (374) 579-3128 | |||
 | wxh5168@126.com wxh5168@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Nanjing Fred Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fredbio.com | |||
 | +86 (25) 8469-6168 | |||
 | Austin@fredchem.cn | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: NJFred01 | |||
 | WhatsApp:+86 17302533743 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Achemica | Switzerland | |||
|---|---|---|---|---|
 | www.achemica.com | |||
 | +41 (24) 466-2929 | |||
 | +41 (24) 466-2800 | |||
 | contact@achemica.com | |||
| Chemical manufacturer since 2010 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Carboxylic acid |
|---|---|
| Name | 2,5-Dibromobenzeneacetic acid |
| Synonyms | (2,5-Dibromophenyl)acetic acid |
| Molecular Structure | 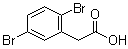 |
| Molecular Formula | C8H6Br2O2 |
| Molecular Weight | 293.94 |
| CAS Registry Number | 203314-28-7 |
| EC Number | 804-900-1 |
| SMILES | C1=CC(=C(C=C1Br)CC(=O)O)Br |
| Density | 2.0±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 376.4±27.0 °C 760 mmHg (Calc.)* |
| Flash point | 181.5±23.7 °C (Calc.)* |
| Index of refraction | 1.625 (Calc.)* |
| Solubility | Very slightly soluble (0.35 g/L water) (25 °C), (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H302-H315-H319-H332-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2,5-Dibromobenzeneacetic acid is an organic compound that consists of a benzene ring substituted with two bromine atoms at the 2 and 5 positions, and an acetic acid group at the 1 position. This compound is part of the broader class of substituted benzene derivatives, which are significant in both organic synthesis and materials science due to their versatility and reactivity. The discovery and development of 2,5-dibromobenzeneacetic acid are situated within the well-established field of halogenated aromatic compounds. The introduction of bromine atoms into the benzene ring often enhances the compound's reactivity and stability, particularly in terms of electrophilic substitution reactions. This specific compound, with its combination of bromine substituents and the carboxylic acid group, has potential utility in the synthesis of other functionalized aromatic compounds, which can be applied in various chemical and industrial processes. One of the major applications of 2,5-dibromobenzeneacetic acid is as a building block in organic synthesis. The bromine atoms in this molecule make it highly reactive in cross-coupling reactions such as Suzuki, Heck, and Sonogashira reactions, where it can form new carbon-carbon bonds with other organic groups. This reactivity is particularly useful in the development of more complex aromatic compounds, which are important in the creation of new materials or pharmaceuticals. The carboxylic acid group at position 1 is also a reactive site, making this molecule suitable for further functionalization or coupling with other molecules through esterification or amidation reactions. In medicinal chemistry, halogenated benzene derivatives like 2,5-dibromobenzeneacetic acid have been explored for their potential pharmacological properties. The presence of bromine atoms can influence the electronic properties of the molecule, which may affect its interaction with biological targets, such as enzymes or receptors. The carboxylic acid group also has the potential to participate in hydrogen bonding or other molecular interactions, which could further enhance the compound's biological activity. Although 2,5-dibromobenzeneacetic acid has not been extensively studied for its specific pharmacological effects, the broader class of halogenated benzene derivatives is known for a wide range of activities, including antimicrobial, anticancer, and anti-inflammatory properties. Furthermore, the molecule may have applications in materials science. The halogen substitution pattern can affect the electronic structure of the molecule, making it useful in the development of electronic materials or sensors. For example, such compounds can be used in organic electronics, such as organic light-emitting diodes (OLEDs) or organic solar cells. The presence of a carboxylic acid group can also make it useful in the formation of self-assembled monolayers or as a precursor to other functional materials. The reactivity of the molecule is also of interest in the field of catalysis. The combination of bromine atoms and the carboxylic acid group can potentially serve as a ligand for metal catalysts, where the compound may facilitate catalytic reactions such as those involved in the formation of complex organic molecules. This could find applications in various industrial processes, including fine chemical synthesis or the production of specialty chemicals. In conclusion, 2,5-dibromobenzeneacetic acid is a valuable intermediate in organic synthesis, with potential applications in materials science, medicinal chemistry, and catalysis. Its halogenated benzene structure provides reactivity that is useful in the creation of more complex molecules, and its carboxylic acid group enables further functionalization or coupling reactions. While its specific biological and industrial applications are still under investigation, it represents a versatile compound that could serve as a starting point for the development of new chemicals or materials. References 2021. An Improved Process for the Enantioselective Synthesis of HCV NS5A Inhibitor Elbasvir (MK-8742) Chiral Amine Intermediate. Russian Journal of General Chemistry, 91(5). DOI: 10.1134/s107036322105025x 2016. Process Chemistry in Antiviral Research. Topics in Current Chemistry, 374(6). DOI: 10.1007/s41061-016-0076-5 2017. Elbasvir. Pharmaceutical Substances, 1. URL: https://www.thieme.de/en/thieme-chemistry/pharmaceutical-substances-54712.htm |
| Market Analysis Reports |