Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Boron Molecular Inc. | USA | |||
|---|---|---|---|---|
 | www.boronmolecular.com | |||
 | +1 (919) 981-4441 | |||
 | +1 (919) 981-4442 | |||
 | sales@boronmolecular-usa.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou TJM Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjmchem.com | |||
 | +86 13388471767 | |||
 | +86 (571) 8822-3279 | |||
 | sales@tjmchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Yancheng Longshen Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.longshenchem.com | |||
 | +86 (515) 8773-1896 +86 18261286666 | |||
 | +86 (515) 8865-2999 | |||
 | 2355756927@qq.com gaojunwyt@vip.163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Oakwood Products, Inc. | USA | |||
|---|---|---|---|---|
 | www.oakwoodchemical.com | |||
 | +1 (800) 467-3386 | |||
 | k_weaver@oakwoodchemical.com | |||
| Chemical manufacturer | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Hydrocarbon halide |
|---|---|
| Name | 1,3-Dibromobenzene |
| Synonyms | M-Dibromobenzene |
| Molecular Structure | 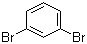 |
| Molecular Formula | C6H4Br2 |
| Molecular Weight | 235.90 |
| CAS Registry Number | 108-36-1 |
| EC Number | 203-574-2 |
| SMILES | C1=CC(=CC(=C1)Br)Br |
| Density | 1.9±0.1 g/cm3, Calc.*, 1.952 g/mL (Expl.) |
|---|---|
| Melting point | -7 °C (Expl.) |
| Index of Refraction | 1.599, Calc.*, 1.608 (Expl.) |
| Boiling Point | 217.5±13.0 °C (760 mmHg), Calc.*, Melting point |
| Flash Point | 93.9 °C, Calc.*, 93 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Transport Information | UN 2711 | ||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
1,3-Dibromobenzene, also known as meta-dibromobenzene, is an aromatic halide with the molecular formula C6H4Br2. It is one of the three positional isomers of dibromobenzene, with the two bromine atoms substituted at the 1 and 3 positions on the benzene ring. The compound appears as a white to off-white crystalline solid at room temperature and has a melting point of approximately 86 °C. It is sparingly soluble in water but readily soluble in organic solvents such as ethanol, ether, and chloroform. The synthesis of 1,3-dibromobenzene was first reported in the late 19th century during systematic studies on the bromination of benzene and its derivatives. Electrophilic aromatic substitution of bromine on benzene in the presence of iron(III) bromide as a catalyst yields bromobenzene, which upon further bromination produces dibrominated isomers. Selective synthesis of the meta isomer requires careful control of reaction conditions and purification steps to separate it from the ortho and para isomers. 1,3-Dibromobenzene serves as a valuable intermediate in organic synthesis, especially in the preparation of more complex aromatic compounds. One of its primary applications is in the field of cross-coupling reactions, such as Suzuki-Miyaura, Stille, and Heck reactions, where it acts as a dihalogenated aryl source. In these reactions, the bromine atoms can be selectively replaced by various functional groups or carbon frameworks, allowing the formation of biaryl structures or substituted aromatics. In material science, 1,3-dibromobenzene is employed as a precursor in the synthesis of conjugated organic molecules and polymers. These materials often exhibit semiconducting or optoelectronic properties and are used in the development of organic light-emitting diodes (OLEDs), organic field-effect transistors (OFETs), and organic photovoltaic devices (OPVs). The symmetrical substitution pattern of the meta isomer provides desirable structural features for tuning molecular geometry and electronic distribution in target compounds. In coordination chemistry, 1,3-dibromobenzene has been used as a ligand precursor or a building block for ligand frameworks. The bromine atoms serve as functional handles for subsequent transformations, such as lithiation or metalation, enabling the synthesis of chelating ligands or organometallic complexes. These complexes are studied for their catalytic activity or electronic properties. In medicinal chemistry, the compound has been utilized in the design and synthesis of pharmacologically active molecules. Although 1,3-dibromobenzene itself is not a drug or therapeutic agent, its brominated aromatic framework is useful in constructing molecules with antimicrobial, anticancer, or anti-inflammatory potential. The bromine atoms can be selectively replaced or functionalized to introduce bioactive moieties. From an environmental and toxicological perspective, 1,3-dibromobenzene is considered hazardous due to its potential to cause irritation to the skin, eyes, and respiratory tract. It should be handled using appropriate safety measures in a well-ventilated area or under a fume hood, with personal protective equipment. It is also subject to regulations concerning halogenated aromatic compounds due to their persistence and potential ecological impact. Overall, 1,3-dibromobenzene is a well-established compound in the chemical literature, with documented uses in synthetic chemistry, materials science, and intermediate production. Its symmetrical substitution and reactivity make it a versatile reagent in various chemical transformations, with applications that span academic and industrial research. References 2024. Mechanochemical Monoarylation of Dihaloarenes Enabled by In-Situ Crystallization. Palladium-Catalyzed Mechanochemical Cross-Coupling Reactions, 183-204. DOI: 10.1007/978-981-97-1991-4_7 2021. Synthesis of Phenols. Science of Synthesis. 1893. Ü ber die Einwirkung von Natrium auf Orthodibrombenzol. Monatshefte für Chemie, 14(1), 517-527. DOI: 10.1007/bf01517879 |
| Market Analysis Reports |