Online Database of Chemicals from Around the World
| Zhengzhou Yuanli Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zzyuanli.cn | |||
 | +86 (0371) 6789-7870 | |||
 | sales08@zzyuanli.cn | |||
 | QQ Chat | |||
 | WeChat: 15137288306 | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Chemical reagent >> Organic reagent >> Borane |
|---|---|
| Name | 1,2-Dicarba-closo-dodecaboran-1-ylacetic acid |
| Molecular Structure | 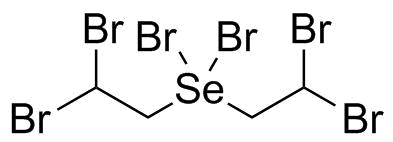 |
| Molecular Formula | B10C4H14O2 |
| Molecular Weight | 202.26 |
| CAS Registry Number | 20644-59-1 |
| SMILES | C(C(Br)Br)[Se](CC(Br)Br)(Br)Br |
|
1,2-Dicarba-closo-dodecaboran-1-ylacetic acid is a boron cluster compound derived from the **carborane** family, specifically based on the 1,2-dicarba-closo-dodecaborane cage (commonly referred to as *o-carborane*). Its structure consists of a highly stable icosahedral cluster composed of ten boron atoms and two carbon atoms, with an acetic acid substituent (–CH2COOH) attached to one of the carbon vertices. This combination of a boron-rich cage and a functional organic group gives the compound unique chemical and physical properties. The molecular structure features a **closo-dodecaborane cage**, an approximately spherical, electron-delocalized cluster with remarkable thermal and chemical stability. The two carbon atoms occupy adjacent positions (1,2-positions) within the cage, and one of these carbons is substituted with an acetic acid side chain. The carboxylic acid group provides a reactive site for further chemical transformations, while the boron cluster contributes hydrophobicity, steric bulk, and resistance to chemical degradation. Synthesis of 1,2-dicarba-closo-dodecaboran-1-ylacetic acid typically involves functionalization of o-carborane through lithiation at the carbon vertex followed by reaction with electrophiles such as haloacetic acid derivatives. Careful control of reaction conditions is required to selectively functionalize the carbon site without disrupting the boron cage structure. The resulting compound retains the integrity of the icosahedral cluster while introducing a versatile carboxylic acid functionality. One of the most significant areas of application for this compound is in medicinal and pharmaceutical chemistry, particularly in **boron neutron capture therapy (BNCT)**. The boron-rich cage allows for high boron content per molecule, which is essential for delivering sufficient boron atoms to tumor cells. The acetic acid group can be further modified to attach targeting moieties, improving selectivity toward specific biological tissues. In addition to biomedical applications, this compound is used in materials science and coordination chemistry. The carboxylic acid group enables incorporation into polymers, metal–organic frameworks, and other advanced materials, while the carborane cage contributes exceptional thermal stability, chemical resistance, and unique electronic properties. These features are valuable in designing high-performance materials, catalysts, and specialty coatings. Overall, 1,2-dicarba-closo-dodecaboran-1-ylacetic acid is a versatile boron cluster compound that combines the extraordinary stability of a carborane cage with the reactivity of a carboxylic acid group. Its high boron content, chemical robustness, and functionalization capability make it important in medicinal chemistry, advanced materials, and specialized synthetic applications. References none |
| Market Analysis Reports |