Online Database of Chemicals from Around the World
| Abosyn Chemicals Inc. | USA | |||
|---|---|---|---|---|
 | www.abosyn.com | |||
 | +1 (203) 683-8328 | |||
 | info@abosyn.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Ketones |
|---|---|
| Name | 3-(4-Bromo-1-oxoisoindolin-2-yl)piperidine-2,6-dione |
| Synonyms | 3-(7-bromo-3-oxo-1H-isoindol-2-yl)piperidine-2,6-dione |
| Molecular Structure | 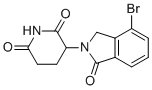 |
| Molecular Formula | C13H11BrN2O3 |
| Molecular Weight | 323.14 |
| CAS Registry Number | 2093387-36-9 |
| SMILES | C1CC(=O)NC(=O)C1N2CC3=C(C2=O)C=CC=C3Br |
| Density | 1.7±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.651, Calc.* |
| Boiling Point | 588.0±50.0 °C (760 mmHg), Calc.* |
| Flash Point | 309.4±30.1 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335-H361 Details |
| Safety Statements | P203-P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P318-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details |
| SDS | Available |
|
The discovery of 3-(4-bromo-1-oxoisoindolin-2-yl)piperidine-2,6-dione (commonly abbreviated as BIPD) stems from the exploration of the promising biological activities of isoindolinone derivatives. Isoindolinone has a rigid bicyclic structure and a lactam ring, which contributes to its stability and potential as a therapeutic agent. By introducing the piperidine-2,6-dione moiety and a bromine atom at the 4-position, researchers developed BIPD to enhance the pharmacological properties and reactivity of the compound. The synthesis of BIPD involves a multistep process that begins with the construction of the isoindolinone core, followed by bromination and the addition of the piperidinedione group. This synthetic route highlights the complexity of the compound and the precision required of modern organic chemistry. BIPD is presented as a crystalline solid, and its chemical stability and unique reactivity are attributed to the presence of the bromine atom and the piperidinedione moiety. BIPD can serve as a scaffold for drug discovery, providing multiple sites for chemical modification. Its isoindolinone core can effectively bind to a variety of biological targets, making it a valuable starting point for the design of new therapeutic agents. Researchers have used BIPD to create analogs and derivatives with improved pharmacokinetic and pharmacodynamic properties. BIPD derivatives have shown potential as anticancer agents. The compound's ability to interfere with cell cycle regulation and induce apoptosis makes it a candidate for the development of drugs targeting cancer cells. The bromine atom enhances the compound's ability to interact with cancer-associated proteins, aiding in preclinical studies. BIPD is used to develop enzyme inhibitors. Its structure allows for the design of compounds that can effectively bind and inhibit enzyme activity, providing potential treatments for enzyme dysfunction diseases such as cancer and metabolic disorders. The piperidinedione moiety contributes to the compound's binding affinity, enhancing its potential as an inhibitor. Research on BIPD derivatives includes its use as an anti-inflammatory agent. The compound's ability to modulate inflammatory pathways offers the possibility of treating diseases such as arthritis and other inflammatory diseases. Modifications to BIPD's structure are designed to improve its selectivity and minimize side effects, making it a promising candidate for the development of new anti-inflammatory drugs. BIPD is a valuable intermediate in organic synthesis. Its isoindolinone core and piperidinedione moiety combination provide a platform for a variety of chemical transformations. The bromine atom promotes cross-coupling reactions, allowing the creation of complex molecular structures necessary for the synthesis of pharmaceuticals and fine chemicals. The compound is used as a building block for the synthesis of heterocyclic compounds. The structural diversity of isoindolinone and piperidinedione rings allows the creation of heterocycles with potential biological activity, which is essential for the development of new compounds for pharmaceuticals and materials science. As a precursor, BIPD can be further functionalized to introduce additional substituents, creating derivatives with tailored properties. This versatility allows the synthesis of a wide range of compounds with specific electronic and steric characteristics for pharmaceuticals and advanced materials. References 2022. A new method for the preparation of 2-(2-(2,6-dioxopiperidin-3-yl)-1-oxoisoindolin-4-yl)acetic acid. CN-115215835-A 2021. Protein inhibitor or degrading agent, pharmaceutical composition containing same and pharmaceutical use. WO-2022268229-A1 2021. Modulators of bcl6 proteolysis and associated methods of use. WO-2022221673-A1 |
| Market Analysis Reports |