Online Database of Chemicals from Around the World
| Shanghai Yurlic Chemical S&T Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yurlichem.com | |||
 | +86 (21) 37285141 +86 15000957566 | |||
 | +86 (21) 37285142 / 5736-5707 | |||
 | yurlichem@hotmail.com sales@yurlichem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Huzhou Zhuorui Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zrchemtech.com | |||
 | +86 189 6805 0179 | |||
 | sales@zrchemtech.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2020 | ||||
| Classification | Organic raw materials >> Organic derivative of hydrazine or hydrazine |
|---|---|
| Name | tert-Butyl 2-methylcarbazate |
| Synonyms | 1,1-Dimethylethyl 1-methylhydrazinecarboxylate; 1-Carbo-tert-butoxy-1-methylhydrazine; N-(tert-Butoxycarbonyl)-N-methylhydrazine; N-Boc-N-methylhydrazine; N-Methylhydrazinecarboxylic acid tert-butyl ester; tert-Butyl 1-methylhydrazinecarboxylate |
| Molecular Structure | 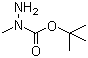 |
| Molecular Formula | C6H14N2O2 |
| Molecular Weight | 146.19 |
| CAS Registry Number | 21075-83-2 |
| EC Number | 629-342-4 |
| SMILES | CC(C)(C)OC(=O)N(C)N |
| Density | 1.029 |
|---|---|
| Boiling point | 186 °C |
| Flash point | 66 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H227 Details | ||||||||||||||||||||||||
| Safety Statements | P501-P210-P280-P370+P378-P403+P235 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Tert-butyl 2-methylcarbazine (Boc-methylhydrazine) is an organic compound with the molecular formula C6H14N2O2. The chemical is primarily used as a protecting group for amines in organic synthesis, particularly in the development of pharmaceuticals and fine chemicals. The discovery and application of tert-butyl 2-methylcarbazine provided a reliable method for protecting and deprotecting functional groups during complex synthetic processes, greatly advancing the field of organic chemistry. The development of tert-butyl 2-methylcarbazine can be linked to the broader exploration of carbazine compounds that began in the mid-20th century when chemists sought to create new reagents for use in synthetic chemistry. The introduction of the tert-butyl group as a protecting group for hydrazines and amines was a particularly useful innovation that allowed for greater control and selectivity in chemical reactions. The tert-butyl group was favored for its stability under a wide range of conditions, which allowed it to protect sensitive functional groups from undesirable side reactions. One of the most important applications of tert-butyl 2-methylcarbazine is in the field of medicinal chemistry. In drug development, it is critical to synthesize complex molecules that often contain multiple functional groups, each of which must be manipulated selectively. tert-Butyl 2-methylcarbazate serves as a protecting group for hydrazine and amine functional groups, allowing chemists to temporarily deactivate these groups while modifying other parts of the molecule. Once the desired transformation is complete, the tert-butyl group can be removed under mild acidic conditions, exposing the free amine or hydrazine for further use or for use in the final product. In addition to its use in drug synthesis, tert-Butyl 2-methylcarbazate is also used to prepare peptide-based therapeutics. Protecting groups are essential in peptide synthesis to prevent unwanted reactions between amino acids. The tert-butyl group helps ensure that the peptide is assembled in the correct order and has the desired structural integrity. This is particularly important for the development of new peptide-based therapeutics that can target specific biological pathways with high precision. tert-Butyl 2-methylcarbazate can also be used in the synthesis of fine chemicals and specialty materials. Its role as a protecting group helps to build complex molecular structures for advanced materials and chemical intermediates. The stability and ease of removal of the tert-butyl group make it an attractive choice for synthetic chemists working in multi-step processes where intermediate compounds must be carefully controlled and manipulated. The versatility of tert-butyl 2-methylcarbazate also extends to academic research. In a laboratory setting, this compound is used to study reaction mechanisms and develop new synthetic methods. Its ability to selectively protect and deprotect functional groups provides a powerful tool for exploring new chemical transformations and optimizing existing processes. Safety and handling are important considerations when working with tert-butyl 2-methylcarbazate. While it is generally stable and non-reactive under normal conditions, appropriate precautions must be taken to avoid exposure and manage any potential risks associated with its use. Proper laboratory protocols and protective equipment are essential to ensure safe handling of this and other chemical reagents. References 2013. Stereodynamics of Nitrogen Chiral Centers in aza-β3-Cyclodipeptides. *Chirality*, 25(9). DOI: 10.1002/chir.22160 2009. Alkylation of Hydrazides. *Science of Synthesis*. URL: SD-040-00831 |
| Market Analysis Reports |