Online Database of Chemicals from Around the World
| Zhejiang Arts & Crafts Imp & Exp.co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjartspharm.com | |||
 | +86 571 87238903 | |||
 | jocelynpan@zjarts.com | |||
| Chemical distributor since 1992 | ||||
| chemBlink Standard supplier since 2024 | ||||
| IOLITEC Ionic Liquids Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.iolitec-usa.com | |||
 | +1 (205) 348-2831 | |||
 | +1 (205) 348-3510 | |||
 | info@iolitec-usa.com | |||
| Chemical manufacturer since 2002 | ||||
| Achemica | Switzerland | |||
|---|---|---|---|---|
 | www.achemica.com | |||
 | +41 (24) 466-2929 | |||
 | +41 (24) 466-2800 | |||
 | contact@achemica.com | |||
| Chemical manufacturer since 2010 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | 1,2-Dimethyl-3-propylimidazolium iodide |
| Synonyms | 1-Propyl-2,3-dimethylimidazolium iodide; 2,3-Dimethylpropylimidazolium iodide; DMPII; N-Methyl-N'-n-propyl-2-methylimidazolium iodide |
| Molecular Structure | 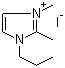 |
| Molecular Formula | C8H15N2.I |
| Molecular Weight | 266.12 |
| CAS Registry Number | 218151-78-1 |
| EC Number | 679-888-2 |
| SMILES | CCCN1C=C[N+](=C1C)C.[I-] |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
1,2-Dimethyl-3-propylimidazolium iodide (DMPI) is a well-known ionic liquid with the chemical formula C₁₀H₁₁N₂I. As a member of the large family of ionic liquids, DMPI has attracted much attention due to its unique properties and potential applications in various scientific and industrial fields. The discovery of DMPI stems from the broader exploration of ionic liquids, which began to gain significant attention in the late 20th century. Researchers sought to develop solvents with unique properties, such as low volatility, high thermal stability, and wide electrochemical windows. DMPI was part of this research, which aimed to explore new ionic liquids with different cation-anion combinations. The synthesis involves the reaction of 1,2-dimethylimidazole with 1-bromopropane, followed by iodide exchange. 1,2-Dimethylimidazole is synthesized from imidazole and a methylating agent, which is then reacted with 1-bromopropane to form 1,2-dimethyl-3-propylimidazolium bromide. 1,2-Dimethyl-3-propylimidazolium bromide is treated with sodium iodide to replace the bromide ions with iodide ions, generating DMPI. DMPI is used in electrochemical applications due to its good ionic conductivity and stability. It can be used as an electrolyte in a variety of electrochemical devices, including batteries and supercapacitors. The high ionic conductivity and thermal stability of DMPI enhance the performance of these devices, making it an important component in energy storage technology. As an ionic liquid, DMPI offers a green alternative to traditional organic solvents. It is non-volatile and has a low environmental impact, making it suitable for use in environmentally friendly processes. DMPI is used in organic synthesis and extraction, helping to improve reaction efficiency and selectivity while minimizing waste and pollution. DMPI is used in catalysis due to its ability to dissolve a wide range of compounds. It acts as both a solvent and a co-catalyst in a variety of catalytic reactions. DMPI's stability and solvation properties make it effective in both homogeneous and heterogeneous catalysis, helping to increase reaction rates and yields. DMPI is very effective in separation processes such as liquid-liquid extraction and ion exchange. Its solvating properties allow for the selective extraction and separation of specific compounds from a mixture. This application is valuable in drug purification and precious metal recovery, improving process efficiency. In materials science, DMPI is used to prepare and modify materials with tailored properties. It plays a role in the synthesis of nanoparticles, polymers, and other advanced materials. DMPI can act as a solvent or stabilizer during the synthesis of nanomaterials, affecting their size, shape, and stability. References 2008. Effects of substituents of imidazolium cations on the performance of dye-sensitized TiO2 solar cells. Journal of Applied Electrochemistry, 38(5). DOI: 10.1007/s10800-008-9611-6 2024. A novel binary electrolyte 1-allyl-3-methylimidazolium dicyanamide ionic liquid/acetonitrile-iodide for sustainable dye-sensitized solar cells. Ionics, 30(10). DOI: 10.1007/s11581-024-05865-y 2004. Novel room temperature ionic liquids of hexaalkyl substituted guanidinium salts for dye-sensitized solar cells. Applied Physics A, 79(4-6). DOI: 10.1007/s00339-003-2505-x |
| Market Analysis Reports |