Online Database of Chemicals from Around the World
| Hunan Yixiang Chemical Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hnyxchem.com.cn | |||
 | +86 (731) 8328-2288 | |||
 | +86 (731) 8328-1399 | |||
 | sales@hnyxchem.com.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Tianjin Icason Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.icason.com | |||
 | +86 (22) 6093-8150 | |||
 | +86 (22) 2562-1236 | |||
 | icason002@icason.com | |||
| Chemical distributor since 1990 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | 1,3-Bis(tert-butylperoxyisopropyl)benzene |
| Synonyms | 1,3-Bis(2-tert-butylperoxy-2-propyl)benzene; m-Bis[2-(tert-butylperoxy)-2-propyl]benzene |
| Molecular Structure | 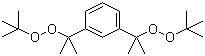 |
| Molecular Formula | C20H34O4 |
| Molecular Weight | 338.48 |
| CAS Registry Number | 2212-81-9 |
| EC Number | 218-664-7 |
| SMILES | CC(C)(C)OOC(C)(C)C1=CC(=CC=C1)C(C)(C)OOC(C)(C)C |
| Density | 1.0±0.1 g/cm3, Calc.*, 0.974 g/cm3 |
|---|---|
| Index of Refraction | 1.475, Calc.* |
| Boiling Point | 360.1±35.0 °C (760 mmHg), Calc.* |
| Flash Point | 129.6±25.8 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS02 Danger Details GHS02 Danger Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H242-H413 Details | ||||||||||||
| Safety Statements | P210-P234-P235-P240-P273-P280-P370+P378-P403-P410-P411-P420-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
|
1,3-Bis(tert-butylperoxyisopropyl)benzene, commonly abbreviated as BTPB, is a chemical compound with significant utility in polymer chemistry, particularly as a radical initiator in polymerization processes. This compound, characterized by its two tert-butylperoxy groups attached to a benzene ring, plays a crucial role in the production of various types of polymers, including rubbers and plastics, due to its ability to efficiently generate free radicals under controlled conditions. The discovery of BTPB dates back to the mid-20th century when researchers were exploring new ways to improve the efficiency of polymerization reactions. The challenge at the time was to find compounds that could initiate polymerization at relatively low temperatures, thereby enhancing the control over the polymerization process and the properties of the resulting polymers. The introduction of peroxides, particularly organic peroxides like BTPB, revolutionized this area of chemistry. The design of BTPB, with its stable tert-butylperoxy groups, provided a reliable source of free radicals, which are essential for initiating the polymerization of monomers into polymers. BTPB is primarily used as a radical initiator in the polymer industry. Its ability to decompose at elevated temperatures to produce free radicals makes it ideal for initiating the polymerization of monomers such as styrene, acrylates, and methacrylates. One of the significant advantages of BTPB is its relatively high thermal stability compared to other peroxides. This stability allows for its use in processes that require precise temperature control, ensuring that the polymerization occurs uniformly and with minimal side reactions. In the production of elastomers, particularly in the vulcanization of rubber, BTPB serves as a cross-linking agent. During vulcanization, BTPB generates free radicals that facilitate the formation of cross-links between polymer chains, enhancing the mechanical strength and elasticity of the rubber. This process is critical in the manufacturing of tires, seals, and various other rubber products where durability and flexibility are required. Beyond its application in polymerization and vulcanization, BTPB is also used in the curing of thermosetting resins. Thermosetting resins, such as unsaturated polyesters and vinyl esters, require the formation of a cross-linked network to achieve their final, hardened state. BTPB is employed in these systems to initiate the curing process, leading to the formation of highly durable materials used in automotive parts, construction materials, and electrical components. The chemical structure of BTPB allows it to be tailored for specific applications by adjusting the reaction conditions under which it is used. For example, by varying the temperature and the concentration of BTPB in a polymerization reaction, chemists can control the molecular weight and distribution of the resulting polymer, as well as its physical properties. This flexibility is particularly important in the development of advanced materials, where precise control over polymer characteristics is necessary to meet specific performance criteria. However, the use of BTPB, like other organic peroxides, requires careful handling due to its potential hazards. BTPB can decompose exothermically, leading to the release of heat and gases, which can pose risks if not properly managed. As a result, strict safety protocols are in place for the storage, transportation, and use of BTPB to prevent accidental decomposition and ensure safe operation in industrial settings. In summary, 1,3-Bis(tert-butylperoxyisopropyl)benzene is a vital compound in the polymer industry, valued for its ability to efficiently initiate polymerization and cross-linking reactions. Its discovery has had a profound impact on the production of a wide range of polymeric materials, contributing to advancements in rubber manufacturing, thermosetting resin curing, and the development of high-performance polymers. As polymer chemistry continues to evolve, BTPB remains an essential tool in the creation of materials that are integral to modern technology and industry. References 2022. Use of Fluoroperoxides Derived from Polyfluorinated Di- and Tetracarbonyl Compounds for Vulcanization of Fluoroelastomers. Russian Journal of Applied Chemistry, 95(10). DOI: 10.1134/s1070427222100020 2017. Polypropylene polymer blends. Russian Journal of General Chemistry, 87(9). DOI: 10.1134/s1070363217090523 1987. Advances in Nitrile Rubber (NBR). Developments in Rubber Technology—4. DOI: 10.1007/978-94-009-3435-1_2 |
| Market Analysis Reports |