Online Database of Chemicals from Around the World
| Mascot I.E. CO.,Ltd. | China | |||
|---|---|---|---|---|
 | www.mascotchem.com | |||
 | +86 (519) 8501-0339 +86 13584504415 | |||
 | +86 (519) 8501-0366 | |||
 | info@mascotchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2006 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Xiamen Grandachem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.grandachem.com | |||
 | +86 (592) 533-3280 | |||
 | +86 (592) 533-3231 | |||
 | info@grandachem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Scientific Polymer Products, Inc. | USA | |||
|---|---|---|---|---|
 | www.scientificpolymer.com | |||
 | +1 (585) 265-0413 | |||
 | +1 (585) 265-1390 | |||
 | custserv@scipoly.com | |||
| Chemical manufacturer | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester >> Acid ester compound |
|---|---|
| Name | Neopentyl glycol diacrylate |
| Synonyms | 2,2-Dimethyl-1,3-propanediyl diacrylate |
| Molecular Structure | 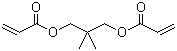 |
| Molecular Formula | C11H16O4 |
| Molecular Weight | 212.24 |
| CAS Registry Number | 2223-82-7 |
| EC Number | 218-741-5 |
| SMILES | CC(C)(COC(=O)C=C)COC(=O)C=C |
| Density | 1.031 |
|---|---|
| Refractive index | 1.453 |
| Flash point | >110 °C |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H310-H311-H315-H317-H319 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P262-P264-P264+P265-P270-P272-P280-P302+P352-P305+P351+P338-P316-P321-P332+P317-P333+P317-P337+P317-P361+P364-P362+P364-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 2810 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Neopentyl glycol diacrylate (NPGDA) is a significant chemical compound in the realm of polymer chemistry, known for its role as a crosslinking agent and a reactive diluent. It is recognized for its unique chemical structure and versatile applications, which contribute to its importance in the synthesis of advanced polymeric materials. The discovery of neopentyl glycol diacrylate can be traced back to the advancements in polymer science during the mid-20th century. Researchers were exploring ways to enhance the properties of polymers and resins by incorporating various functional groups that could improve their mechanical and chemical properties. The development of NPGDA arose from the need for crosslinking agents that could provide high reactivity and stability, which are crucial for achieving desirable characteristics in polymeric materials. Neopentyl glycol diacrylate is synthesized from neopentyl glycol and acrylic acid. The chemical structure of NPGDA consists of two acrylate groups attached to a neopentyl glycol backbone. This structure endows NPGDA with a high reactivity towards polymerization processes, particularly through free radical mechanisms. The presence of two acrylate functional groups allows for extensive crosslinking within polymer matrices, leading to the formation of robust and durable polymer networks. One of the primary applications of NPGDA is in the formulation of UV-curable coatings and inks. In these applications, NPGDA serves as a reactive diluent that not only reduces the viscosity of the formulation but also contributes to the crosslinking density of the cured film. When exposed to UV light, NPGDA undergoes polymerization, leading to the formation of a hardened, crosslinked network. This property is highly valued in the coatings industry, where UV-cured coatings offer rapid curing times, excellent chemical resistance, and superior durability. In addition to coatings, NPGDA is extensively used in the production of adhesives and sealants. The crosslinking capability of NPGDA enhances the mechanical strength and adhesive properties of these materials. By incorporating NPGDA into adhesive formulations, manufacturers can achieve stronger bonds and improve the overall performance of the adhesive under various environmental conditions. Similarly, in sealants, NPGDA contributes to the formation of a resilient, long-lasting seal that can withstand exposure to moisture and chemicals. The use of NPGDA extends to the field of dental materials as well. In dental composites and resins, NPGDA is employed as a crosslinking agent to improve the mechanical properties and longevity of dental restorations. The ability of NPGDA to form a stable, crosslinked network ensures that dental materials can endure the stresses and strains encountered in the oral environment, thereby enhancing the durability of dental restorations. Another notable application of NPGDA is in the development of high-performance polymers and composites. The crosslinking provided by NPGDA leads to polymers with enhanced thermal stability, mechanical strength, and chemical resistance. These properties make NPGDA-containing polymers suitable for demanding applications in industries such as automotive, aerospace, and electronics. Despite its advantages, the use of NPGDA requires careful handling due to its reactive nature. The polymerization of NPGDA can generate heat, and appropriate measures must be taken to control the reaction conditions and prevent unwanted side reactions. Additionally, as with other acrylate-based compounds, safety precautions are necessary to mitigate potential health risks associated with exposure to NPGDA and its derivatives. In conclusion, neopentyl glycol diacrylate is a valuable chemical substance with diverse applications in polymer chemistry. Its discovery and subsequent utilization in various industrial processes have significantly advanced the development of high-performance coatings, adhesives, dental materials, and polymers. The unique properties of NPGDA, including its ability to facilitate crosslinking and improve material performance, continue to make it an essential component in the production of advanced materials across multiple industries. References 2024. Hydrolytic degradation and biomineralization of amine-cured epoxy resin based on glycidate. Polymer Journal, 56(3). DOI: 10.1038/s41428-023-00880-8 2022. Fabrication of anti-counterfeiting patterns with angle-dependent colors by silkscreen printing and UV-curable photonic crystal inks. Science China Materials, 66(4). DOI: 10.1007/s40843-022-2256-8 2006. Chemistry of Photocurable Compositions. Light-Associated Reactions of Synthetic Polymers. DOI: 10.1007/0-387-36414-5_3 |
| Market Analysis Reports |