Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Cn Chemunion Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cnchemunion.com | |||
 | +86 17366116869 | |||
 | david@cnchemunion.com | |||
 | QQ Chat | |||
 | WeChat: +86 15250069576 | |||
 | WhatsApp:+86 17366116869 | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Hormone and endocrine-regulating drugs >> Pancreatic hormones and other blood sugar regulating drugs |
|---|---|
| Name | Danuglipron |
| Synonyms | 2-[[4-[6-[(4-cyano-2-fluorophenyl)methoxy]pyridin-2-yl]piperidin-1-yl]methyl]-3-[[(2S)-oxetan-2-yl]methyl]benzimidazole-5-carboxylic acid |
| Molecular Structure | 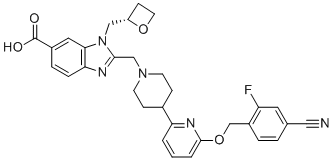 |
| Molecular Formula | C31H30FN5O4 |
| Molecular Weight | 555.60 |
| CAS Registry Number | 2230198-02-2 |
| SMILES | C1CO[C@@H]1CN2C3=C(C=CC(=C3)C(=O)O)N=C2CN4CCC(CC4)C5=NC(=CC=C5)OCC6=C(C=C(C=C6)C#N)F |
| Density | 1.4±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.680, Calc.* |
| Boiling Point | 782.6±60.0 °C (760 mmHg), Calc.* |
| Flash Point | 427.1±32.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
| SDS | Available |
|
Danuglipron is a novel investigational compound primarily developed for the treatment of type 2 diabetes. It acts as an agonist for the glucagon-like peptide-1 receptor (GLP-1R), a key target in glucose regulation. The discovery of danuglipron was part of the ongoing efforts to create more effective and convenient therapies for managing blood sugar levels in individuals with diabetes. GLP-1R agonists have gained significant attention due to their ability to stimulate insulin secretion, inhibit glucagon release, and slow gastric emptying, all of which contribute to improved glycemic control. Danuglipron is notable for its oral bioavailability, which represents a significant advancement over traditional GLP-1R agonists that are typically administered via injection. This oral formulation offers greater convenience and improved patient adherence to treatment regimens. Its pharmacokinetic profile allows for steady and prolonged activity, helping to maintain stable blood glucose levels throughout the day. The compound has shown promise in clinical trials, demonstrating effectiveness in reducing glycated hemoglobin (HbA1c) levels and promoting weight loss, both crucial factors in diabetes management. The development of danuglipron addresses a growing need for oral diabetes medications that provide the benefits of GLP-1R agonists without the need for frequent injections. Its mechanism of action involves mimicking the effects of endogenous GLP-1, a hormone released in response to food intake that helps regulate blood sugar levels. By activating the GLP-1 receptor, danuglipron enhances insulin secretion in response to meals while simultaneously reducing excessive glucagon secretion, which contributes to high blood sugar levels in diabetic patients. Danuglipron has also shown potential applications beyond diabetes treatment. Research is exploring its use in weight management, as GLP-1R agonists are known to suppress appetite and reduce food intake. This makes the compound a candidate for addressing obesity, a common comorbidity in patients with type 2 diabetes. Its oral administration and favorable safety profile could make it an attractive option for long-term use in both diabetes and obesity management. The discovery and ongoing development of danuglipron reflect the pharmaceutical industry's commitment to advancing diabetes care through innovative therapies that offer both efficacy and convenience. As research continues, danuglipron may emerge as a key player in the future of diabetes treatment, providing patients with a highly effective and user-friendly option for managing their condition. References 2022. A Small-Molecule Oral Agonist of the Human Glucagon-like Peptide-1 Receptor. Journal of Medicinal Chemistry, 65(12). DOI: 10.1021/acs.jmedchem.1c01856 2022. A phase 1 study to evaluate the safety, tolerability, pharmacokinetics and pharmacodynamics of danuglipron (PF-06882961), an oral small-molecule glucagon-like peptide-1 receptor agonist, in Japanese adults with type 2 diabetes mellitus. Diabetes, Obesity and Metabolism, 25(3). DOI: 10.1111/dom.14928 2021. Danuglipron (PF-06882961) in type 2 diabetes: a randomized, placebo-controlled, multiple ascending-dose phase 1 trial. Nature Medicine, 27(6). DOI: 10.1038/s41591-021-01391-w |
| Market Analysis Reports |