Online Database of Chemicals from Around the World
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Piperidines |
|---|---|
| Name | 1-(4-Nitrophenyl)piperidine-4-carboxylic acid |
| Molecular Structure | 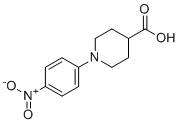 |
| Molecular Formula | C12H14N2O4 |
| Molecular Weight | 250.25 |
| CAS Registry Number | 223786-53-6 |
| SMILES | C1CN(CCC1C(=O)O)C2=CC=C(C=C2)[N+](=O)[O-] |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P280-P301+P312-P302+P352-P304+P340-P305+P351+P338 Details |
| SDS | Available |
|
1-(4-Nitrophenyl)piperidine-4-carboxylic acid is a chemical compound of considerable interest in medicinal chemistry and organic synthesis. This substance combines a piperidine ring with a nitrophenyl group and a carboxylic acid moiety, making it a versatile building block for various chemical applications. The discovery of 1-(4-Nitrophenyl)piperidine-4-carboxylic acid was driven by its potential applications in pharmaceutical chemistry. The compound's structure includes a piperidine ring, which is a common motif in drug design due to its ability to interact with biological targets. The nitrophenyl group serves as a functional handle for further chemical modifications, while the carboxylic acid group facilitates its use in various reactions. In terms of applications, 1-(4-Nitrophenyl)piperidine-4-carboxylic acid is primarily used as an intermediate in the synthesis of pharmaceuticals and other bioactive molecules. Its ability to undergo further chemical transformations makes it a valuable precursor in the development of complex drug candidates. For example, it can be utilized in the synthesis of compounds with potential therapeutic properties, including those targeting central nervous system disorders. The compound’s utility extends beyond pharmaceuticals. Its structural features also make it a useful reagent in organic synthesis for creating other complex molecules. The piperidine ring's versatility allows for the introduction of various substituents, while the nitrophenyl group provides opportunities for further functionalization. Overall, 1-(4-Nitrophenyl)piperidine-4-carboxylic acid represents a significant advancement in chemical synthesis. Its role as a versatile intermediate underscores its importance in both drug development and organic chemistry. The compound's ability to participate in diverse chemical reactions makes it a valuable tool for researchers and chemists aiming to create new and innovative molecules. References 2024. In Silico Design, Synthesis, and Evaluation of PROTAC Against Hematopoietic Prostaglandin D Synthase. *Methods in Molecular Biology (Clifton, N.J.)*. DOI: 10.1007/978-1-0716-3985-6_18 |
| Market Analysis Reports |