Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| SobhaBio | India | |||
|---|---|---|---|---|
 | www.sobhabio.com | |||
 | +91 (99) 8988-2177 | |||
 | sobhabio@gmail.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Classification | Biochemical >> Plant extracts |
|---|---|
| Name | 7-Methoxyflavone |
| Synonyms | 7-Methoxy-2-phenyl-4H-benzopyran-4-one |
| Molecular Structure | 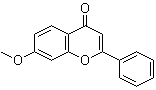 |
| Molecular Formula | C16H12O3 |
| Molecular Weight | 252.26 |
| CAS Registry Number | 22395-22-8 |
| SMILES | COC1=CC2=C(C=C1)C(=O)C=C(O2)C3=CC=CC=C3 |
| Density | 1.2$+/-$0.1 g/cm3 Calc.*, 1.24 g/mL (Expl.) |
|---|---|
| Melting point | 108 - 112 $degree$C (Expl.) |
| Boiling point | 421.2$+/-$45.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 200.3$+/-$15.1 $degree$C (Calc.)* |
| Index of refraction | 1.614 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
7-Methoxyflavone is a methoxylated flavone belonging to the flavonoid family of polyphenolic compounds. It possesses the characteristic flavone skeleton consisting of a benzopyranone core linked to a phenyl ring at the 2-position. In this compound, a methoxy group (–OCH3) is attached at the 7-position of the flavone A-ring, giving rise to a monomethoxylated flavone structure. The molecular framework of 7-methoxyflavone is based on the flavone nucleus, also known as 2-phenyl-4H-1-benzopyran-4-one. This conjugated aromatic system contains a pyrone ring fused to a benzene ring and connected to an additional phenyl substituent. The extended π-electron conjugation contributes to the compound’s ultraviolet absorption properties and aromatic stability. The methoxy substituent at the 7-position influences the electronic and physicochemical properties of the molecule. Methoxy groups are electron-donating through resonance interactions, increasing electron density within the aromatic ring system. Compared with hydroxyflavones, methoxylated flavones generally exhibit increased lipophilicity and altered metabolic behavior due to replacement of hydrogen-bond-donating hydroxyl groups with methyl ether functionality. 7-Methoxyflavone has been identified in natural product studies involving certain plants and botanical extracts. Flavones are biosynthesized in plants through the phenylpropanoid and polyketide pathways. Chalcone intermediates undergo cyclization and oxidation to form the flavone skeleton, after which O-methyltransferase enzymes can methylate hydroxyl groups to produce methoxy-substituted flavones such as 7-methoxyflavone. The compound has attracted interest in natural products chemistry and pharmacological research because flavones are associated with a broad range of biochemical and biological activities. Investigations involving 7-methoxyflavone have examined its interactions with enzymes, receptors, inflammatory mediators, and oxidative processes as part of broader flavonoid structure–activity studies. Chemically, 7-methoxyflavone exhibits reactivity typical of aromatic methoxy-substituted flavones. The carbonyl group within the pyrone ring participates in conjugation across the molecule and may undergo nucleophilic addition or reduction reactions under suitable conditions. The methoxy group can undergo demethylation to generate the corresponding hydroxyflavone derivative. Synthetic preparation of 7-methoxyflavone commonly involves cyclization of appropriately substituted chalcone intermediates using classical flavone synthesis methods such as the Baker–Venkataraman rearrangement or oxidative cyclodehydration reactions. It can also be obtained by methylation of 7-hydroxyflavone using methylating agents under basic conditions. From a physicochemical standpoint, 7-methoxyflavone is relatively hydrophobic because it contains only one methoxy substituent and lacks strongly ionizable groups. The planar aromatic structure promotes π–π stacking interactions and contributes to its crystallization behavior and spectroscopic properties. The compound absorbs ultraviolet light strongly due to its conjugated aromatic system, a feature common to flavones and related flavonoids. This property has made flavones useful subjects in spectroscopic and photochemical studies. Historically, methoxylated flavones such as 7-methoxyflavone have been important in phytochemistry because their substitution patterns help distinguish plant metabolites and provide insight into biosynthetic pathways. Comparative studies of hydroxy- and methoxy-substituted flavones have also contributed to understanding how structural modifications influence physicochemical and biological properties. Overall, 7-methoxyflavone is a monomethoxylated flavone characterized by a conjugated benzopyranone core and a methoxy substituent at the 7-position. Its aromatic structure, biosynthetic origin, and chemical properties make it a representative member of the flavonoid class widely studied in natural products chemistry and medicinal chemistry. References 2025. In Vitro Evaluation of Flavonoids for Enzyme Inhibition, Antioxidant, Antimicrobial and Anticancer Properties with Molecular Docking Insights. Food Biophysics. DOI: 10.1007/s11483-025-10034-3 2025. Structure activity relationship of flavonoids as PPARγ agonists. Phytochemistry Reviews. DOI: 10.1007/s11101-025-10121-w |
| Market Analysis Reports |