Online Database of Chemicals from Around the World
| Futoh Chemicals Co., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.futoh.co.jp | |||
 | +81 (52) 521-8171 | |||
 | +81 (52) 521-8793 | |||
 | tokoro@futoh.co.jp | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Beijing Guofang Dingsheng Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.gfdschem.com | |||
 | +86 (10) 8904-9254 5909-2859 +86 18610472091 +86 18031679351 | |||
 | +86 (10) 5909-2859 | |||
 | gfdskj@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Xinxiang Runyu Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runvmat.com | |||
 | +86 (373) 775-9608 | |||
 | +86 (373) 775-9608 | |||
 | sales@runvmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Dalchem | Russia | |||
|---|---|---|---|---|
 | www.dalchem.com | |||
 | +7 (8312) 753-772 | |||
 | +7 (8312) 750-799 | |||
 | dregichy@dalchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | 2-(Di-tert-butylphosphino)biphenyl |
| Synonyms | (2-Biphenyl)di-tert-butylphosphine |
| Molecular Structure | 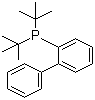 |
| Molecular Formula | C20H27P |
| Molecular Weight | 298.40 |
| CAS Registry Number | 224311-51-7 |
| EC Number | 607-074-9 |
| SMILES | CC(C)(C)P(C1=CC=CC=C1C2=CC=CC=C2)C(C)(C)C |
| Melting point | 84-87 °C |
|---|---|
| Water solubility | Insoluble |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335-H413 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2-(Di-tert-butylphosphino)biphenyl, abbreviated as dtbpf, is a significant compound in organometallic chemistry known for its role as a ligand in transition metal catalysis. The discovery of dtbpf and its applications have markedly impacted various chemical processes, enhancing their efficiency and selectivity. The compound 2-(Di-tert-butylphosphino)biphenyl was first introduced in the early 1990s. Researchers sought to develop new ligands to improve the performance of transition metal catalysts. The structure of dtbpf features a biphenyl backbone with a di-tert-butylphosphino group attached to one of the phenyl rings. This design was intended to optimize both the steric and electronic properties of the ligand, thereby improving its effectiveness in catalysis. The synthesis of dtbpf involves the reaction between 2-biphenyl and di-tert-butylphosphine. The reaction conditions are carefully controlled to ensure a high yield and purity of the final product. This typically requires the use of specific bases and solvents that facilitate the formation of the phosphine-biphenyl linkage while minimizing side reactions. The precise handling of these reaction conditions is crucial for obtaining the desired ligand. 2-(Di-tert-butylphosphino)biphenyl is predominantly used as a ligand in various transition metal-catalyzed reactions. One of its main applications is in cross-coupling reactions, such as the Suzuki-Miyaura and Heck reactions. In these processes, dtbpf facilitates the formation of carbon-carbon bonds. Its bulky di-tert-butylphosphino groups create a unique steric environment that enhances the reactivity and selectivity of the metal centers it coordinates with. This leads to higher yields and selectivity in the synthesis of complex organic molecules. In addition to cross-coupling reactions, dtbpf is also utilized in hydrogenation reactions. The ligand improves the performance of metal catalysts used to reduce unsaturated compounds. By enhancing the activity of these catalysts, dtbpf contributes to higher conversion rates and selectivity in hydrogenation processes. Another significant application of dtbpf is in olefin polymerization. The ligand-based catalysts have shown promise in producing polymers with specific properties. The ability of dtbpf to control the polymerization process allows for the creation of polymers with desired molecular weights and structural attributes. The use of 2-(Di-tert-butylphosphino)biphenyl provides several advantages in catalytic processes. Its steric bulk helps stabilize intermediates and transition states, leading to improved catalytic activity and selectivity. The enhanced performance of dtbpf-based catalysts makes them valuable tools in a variety of chemical transformations. However, there are challenges associated with using dtbpf. The bulky nature of the ligand can sometimes lead to issues such as catalyst deactivation and the formation of by-products. These challenges require ongoing research to address. Scientists are continuously exploring modifications to the ligand's structure or developing alternative ligands to overcome these limitations and improve overall catalyst performance. Future research into 2-(Di-tert-butylphosphino)biphenyl aims to expand its applications and address its limitations. Researchers may focus on optimizing the ligand's structure to enhance its performance in new catalytic processes or developing novel derivatives with improved properties. This ongoing investigation is crucial for advancing the use of dtbpf in various chemical applications and achieving more efficient and selective catalytic processes. References 2021. Palladium-catalyzed amino group arylation of 1,3-disubstituted 1H-pyrazol-5-amine based on Buchwald-Hartwig reaction. *Chemistry of Heterocyclic Compounds*, 57(6). DOI: 10.1007/s10593-021-02961-z 2015. Synthesis of Novel 5-(N-Substituted-Anilino)-8-Hydroxyquinolines via Hartwig-Buchwald Amination Reaction. *Journal of Chemical Sciences*, 127(11). DOI: 10.1007/s12039-015-0960-5 2003. Synthesis of substituted oxindoles from alpha-chloroacetanilides via palladium-catalyzed C[bond]H functionalization. *Journal of the American Chemical Society*, 125(37). DOI: 10.1021/ja037546g |
| Market Analysis Reports |