Online Database of Chemicals from Around the World
| Sayibo Inc. | USA | |||
|---|---|---|---|---|
 | www.sayibo.com | |||
 | +1 (631) 709-8181 | |||
 | +1 (631) 709-8182 | |||
 | info@sayibo.com | |||
| Chemical distributor since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Anhui Royal Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.royal-chem.com | |||
 | +86 (25) 8663-9253 | |||
 | +86 (25) 5227-9193 | |||
 | zoey.zhao@royal-chem.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Ethylene glycol di(3-mercaptopropionate) |
| Synonyms | Ethylene glycol bis(beta-mercaptopropionate); Ethylene glycol di(3-mercaptopropionate); Ethylene glycol di(beta-mercaptopropionate); Ethylenebis(3-mercaptopropionate); Ethylenebis(beta-mercaptopropionate); GDMP; Glycol dimercaptopropionate; Thiocure GDMP |
| Molecular Structure | 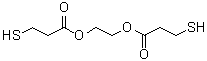 |
| Molecular Formula | C8H14O4S2 |
| Molecular Weight | 238.32 |
| CAS Registry Number | 22504-50-3 |
| EC Number | 245-044-3 |
| SMILES | C(CS)C(=O)OCCOC(=O)CCS |
| Solubility | Slightly soluble (1.4 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.218±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 148 °C** |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2016 ACD/Labs) |
| ** | Klarer, Joseph |
| Hazard Symbols |    GHS05;GHS07;GHS09 Danger Details GHS05;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H312-H302-H312-H317-H318-H319-H400-H410-H411 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P272-P273-P280-P301+P317-P302+P352-P305+P351+P338-P305+P354+P338-P317-P321-P330-P333+P317-P337+P317-P362+P364-P391-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 3082 | ||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
Ethylene glycol di(3-mercaptopropionate) is a notable chemical compound in the field of polymer chemistry, recognized for its role as a crosslinking agent and a reactive component in various polymeric systems. This compound has been pivotal in advancing the properties and applications of synthetic polymers and materials. The discovery of ethylene glycol di(3-mercaptopropionate) emerged from research focused on developing new crosslinking agents to enhance the mechanical and chemical properties of polymers. Researchers were particularly interested in compounds that could introduce thiol groups into polymer networks, which are known for their ability to form disulfide bonds and crosslinking structures. Ethylene glycol di(3-mercaptopropionate) was synthesized as part of these investigations, providing a new tool for polymer scientists to control and improve polymer properties. Ethylene glycol di(3-mercaptopropionate) is synthesized through the reaction of ethylene glycol with 3-mercaptopropionic acid. The chemical structure of this compound features two mercaptopropionate groups attached to an ethylene glycol backbone. The presence of the thiol groups (–SH) in its structure is crucial for its role as a crosslinking agent. These thiol groups are highly reactive and can form disulfide bonds under certain conditions, which facilitates the crosslinking process in polymer matrices. One of the primary applications of ethylene glycol di(3-mercaptopropionate) is in the production of thiol-ene and thiol-acrylate resins. In these systems, the compound acts as a crosslinker that helps to form a three-dimensional network of polymer chains. The thiol groups react with double bonds in the polymer backbone, resulting in a crosslinked structure that imparts enhanced mechanical strength, chemical resistance, and thermal stability to the material. Thiol-ene and thiol-acrylate resins are widely used in coatings, adhesives, and composites due to their superior performance characteristics. Ethylene glycol di(3-mercaptopropionate) is also utilized in the development of biomedical materials. In this context, the compound is employed to create hydrogels and other polymeric systems with controlled properties. The ability to crosslink hydrogels using thiol groups allows for the development of materials with specific mechanical properties and swelling behavior, making them suitable for applications in drug delivery, tissue engineering, and wound healing. The biocompatibility of these materials is a critical factor, and ethylene glycol di(3-mercaptopropionate) contributes to achieving the desired performance in biomedical applications. In the field of material science, ethylene glycol di(3-mercaptopropionate) is used to enhance the properties of polymers in various industrial applications. For example, in the manufacture of high-performance composites, the crosslinking provided by this compound improves the overall strength and durability of the material. This is particularly valuable in applications where materials are subjected to harsh environmental conditions or mechanical stresses. Despite its advantages, the use of ethylene glycol di(3-mercaptopropionate) requires careful handling due to its reactive nature. The thiol groups can be prone to oxidation and may react with other substances, necessitating proper storage and usage conditions. Safety precautions are essential to prevent potential health risks associated with exposure to thiol-containing compounds. In summary, ethylene glycol di(3-mercaptopropionate) is an important chemical substance with diverse applications in polymer chemistry. Its role as a crosslinking agent has significantly contributed to the development of advanced materials with enhanced properties. From industrial coatings and adhesives to biomedical materials, the applications of ethylene glycol di(3-mercaptopropionate) highlight its versatility and value in various fields. References 2022. Effect of surfactant concentration and monomer polarity on particle nucleation in emulsion step polymerization of dithiol with diene. *Colloid and Polymer Science*, 300(7). DOI: 10.1007/s00396-022-04993-z 2015. Microwave-Assisted Cationic Ring-Opening Polymerization of 2-Oxazolines. *Advances in Polymer Science*. DOI: 10.1007/12_2015_340 |
| Market Analysis Reports |