Online Database of Chemicals from Around the World
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Bisphenol A bis(2-hydroxyethyl ether) dimethacrylate |
| Synonyms | Diacryl; 2-[4-[2-[4-[2-(2-methylprop-2-enoyloxy)ethoxy]phenyl]propan-2-yl]phenoxy]ethyl 2-methylprop-2-enoate |
| Molecular Structure | 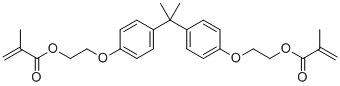 |
| Molecular Formula | C27H32O6 |
| Molecular Weight | 452.54 |
| CAS Registry Number | 24448-20-2 |
| EC Number | 246-263-7 |
| SMILES | CC(=C)C(=O)OCCOC1=CC=C(C=C1)C(C)(C)C2=CC=C(C=C2)OCCOC(=O)C(=C)C |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.527, Calc.* |
| Boiling Point | 574.6±50.0 °C (760 mmHg), Calc.* |
| Flash Point | 243.9±30.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H317-H319-H334-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P233-P260-P261-P264-P264+P265-P271-P272-P280-P284-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P333+P317-P337+P317-P342+P316-P362+P364-P403-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Bisphenol A bis(2-hydroxyethyl ether) dimethacrylate (Bisphenol A BHE-DMA) is a chemical compound widely used in the field of polymer chemistry, particularly in the formulation of high-performance resins and materials. This compound, also known as bisphenol A glycidyl dimethacrylate, features two methacrylate groups attached to a bisphenol A backbone through 2-hydroxyethyl ether linkages. The synthesis of Bisphenol A BHE-DMA involves the reaction of bisphenol A with 2-hydroxyethyl methacrylate, resulting in a compound with two methacrylate ester groups. These groups provide the material with the ability to undergo polymerization under UV light or through free-radical initiation. This property makes Bisphenol A BHE-DMA a valuable component in the production of various high-performance polymers and composites. One of the primary applications of Bisphenol A BHE-DMA is in the development of dental materials. Its excellent mechanical properties, combined with good adhesion to tooth structure, make it ideal for use in dental resins, including composites and adhesives. The compound's ability to form strong, durable bonds with both the dental enamel and the composite materials contributes to its widespread use in restorative dentistry. In the field of coatings, Bisphenol A BHE-DMA is used to produce UV-curable coatings. The presence of methacrylate groups allows for rapid curing under UV light, leading to coatings with high hardness, chemical resistance, and durability. These coatings find applications in a variety of industries, including automotive, electronics, and industrial finishes, where high-performance and long-lasting protection are essential. Furthermore, Bisphenol A BHE-DMA is utilized in the production of high-performance composites. The compound's chemical structure allows it to crosslink effectively, resulting in composites with enhanced mechanical properties and thermal stability. These composites are used in demanding applications such as aerospace components, sporting equipment, and advanced manufacturing processes. In addition to its applications, Bisphenol A BHE-DMA is subject to rigorous safety evaluations. Proper handling and usage are required to ensure safety, given the reactive nature of the methacrylate groups. Adequate protective measures and ventilation are essential to minimize exposure during handling and processing. The versatility and performance characteristics of Bisphenol A BHE-DMA underscore its importance in modern material science. Its use in dental materials, coatings, and composites highlights its role in advancing the functionality and durability of a wide range of products. References 2023. Modern Approaches to Obtaining Organofunctional Silsesquioxanes. *Polymer Science, Series C*, 65(2). DOI: 10.1134/s181123822370039x 2019. Acrylic Resins. *Kanerva’s Occupational Dermatology*. DOI: 10.1007/978-3-319-68617-2_50 2014. Replacing HEMA with alternative dimethacrylates in dental adhesive systems: evaluation of polymerization kinetics and physicochemical properties. *The Journal of Adhesive Dentistry*, 16(3). DOI: 10.3290/j.jad.a31811 |
| Market Analysis Reports |