Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| VanDeMark Chemical Inc. | USA | |||
|---|---|---|---|---|
 | www.vandemark.com | |||
 | +1 (716) 433-6764 | |||
 | +1 (716) 433-2850 | |||
 | sales@vandemark.com | |||
| Chemical manufacturer since 1951 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Cn Chemunion Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cnchemunion.com | |||
 | +86 17366116869 | |||
 | david@cnchemunion.com | |||
 | QQ Chat | |||
 | WeChat: +86 15250069576 | |||
 | WhatsApp:+86 17366116869 | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester |
|---|---|
| Name | 2-Ethylhexyl chloroformate |
| Synonyms | 2-etylhexyl-chlorformiat |
| Molecular Structure | 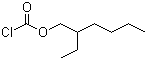 |
| Molecular Formula | C9H17ClO2 |
| Molecular Weight | 192.68 |
| CAS Registry Number | 24468-13-1 |
| EC Number | 246-278-9 |
| SMILES | CCCCC(CC)COC(=O)Cl |
| Density | 0.981 |
|---|---|
| Boiling point | 106-107 °C |
| Refractive index | 1.431 |
| Flash point | 179 °F |
| Hazard Symbols |    GHS05;GHS06;GHS07 Danger Details GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H290-H315-H317-H330-H332 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P234-P260-P261-P264-P271-P272-P280-P284-P302+P352-P304+P340-P316-P317-P320-P321-P332+P317-P333+P317-P362+P364-P390-P403+P233-P405-P406-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2748 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
2-Ethylhexyl chloroformate is an organic chemical compound with the molecular formula C9H17ClO2. It belongs to the class of chloroformates, which are widely used as intermediates in organic synthesis. This compound is typically a colorless liquid and is known for its reactivity, particularly in esterification and urethane formation reactions. The presence of both the chloroformate functional group and the 2-ethylhexyl chain provides versatility in various industrial and laboratory applications. The discovery and synthesis of chloroformates, including 2-ethylhexyl chloroformate, originated from the need to develop reagents that could effectively introduce protective groups or modify chemical structures in a controlled manner. Chloroformates are useful due to their ability to react with alcohols and amines, forming esters and carbamates, respectively. 2-Ethylhexyl chloroformate, in particular, benefits from its branched alkyl chain, which influences its solubility and reactivity. One of the primary applications of 2-ethylhexyl chloroformate is in the production of plasticizers and additives for polymers. Plasticizers improve the flexibility and durability of plastics, and 2-ethylhexyl chloroformate plays a key role in synthesizing compounds that are incorporated into these materials. Its ability to form stable urethanes and esters makes it a valuable intermediate in the chemical processes used to enhance the performance of polymeric products. In addition to its role in polymer chemistry, 2-ethylhexyl chloroformate is used in the pharmaceutical and agrochemical industries. It is often employed in the synthesis of active pharmaceutical ingredients (APIs) and various agricultural chemicals. The reactivity of the chloroformate group allows for the creation of complex molecules with specific biological activities, making it a versatile reagent for drug development and crop protection. 2-Ethylhexyl chloroformate is also used in the preparation of surfactants and coatings. Surfactants are essential in detergents, emulsifiers, and dispersing agents, while coatings derived from 2-ethylhexyl chloroformate provide protective layers for various materials. These applications benefit from the compound’s ability to form stable bonds and impart desirable physical properties to the final product. References 2018. Preparation, Crystal Structures and Hirshfeld Surface Analyses of Two Impurities of Azilsartan. *Journal of Chemical Crystallography*, 48(12). DOI: 10.1007/s10870-018-0754-y 2007. Decarboxylation of Chloroformates. *Science of Synthesis*. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-035-00056 1982. Determination of the components of waste water from bis(2-ethylhexyl)peroxydicarbonate production by gas-solid chromatography. *Fresenius' Zeitschrift für Analytische Chemie*, 311(1). DOI: 10.1007/bf00483717 |
| Market Analysis Reports |