Online Database of Chemicals from Around the World
| Nanjing Search Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.searchbio.com.cn | |||
 | +86 (25) 8168-2922 8586-0978 +86 18913919581 | |||
 | +86 (25) 8586-0962 | |||
 | trade@searchbio.com.cn sales@searchbio.com.cn Linda@searchbio.com.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Extrasynthese Chemical S.A.S. | France | |||
|---|---|---|---|---|
 | www.extrasynthese.com | |||
 | +33 (47) 898-2034 | |||
 | +33 (47) 898-1945 | |||
 | info@extrasynthese.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| SRC Laboratories Pvt Ltd | India | |||
|---|---|---|---|---|
 | www.srclaboratories.com | |||
 | +91 (40) 2307-7999 | |||
 | +91 (40) 2307-7999 | |||
 | info@srclaboratories.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Nanjing Ultra Pure Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ultrapurebio.com | |||
 | +86 (25) 8226-3158 | |||
 | +86 (25) 8226-3168 | |||
 | sales@ultrapurebio.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Real-Times (Beijing) Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.real-times.com.cn | |||
 | +86 (10) 8259-8075 +86 13426307855 | |||
 | +86 (10) 8259-7807 | |||
 | real-times@163.com | |||
| Chemical manufacturer since 2006 | ||||
| Syn-Finechem Laboratories Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.synfinechem.com | |||
 | +91 (40) 2309-5094 | |||
 | +91 (40) 2319-4700 | |||
 | synfine@synfinechem.com info@synfinechem.com | |||
| Chemical manufacturer since 2003 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic ketone |
|---|---|
| Name | Acetosyringone |
| Synonyms | 4'-Hydroxy-3',5'-dimethoxyacetophenone; 3',5'-Dimethoxy-4'-hydroxyacetophenone |
| Molecular Structure | 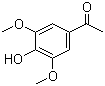 |
| Molecular Formula | C10H12O4 |
| Molecular Weight | 196.20 |
| CAS Registry Number | 2478-38-8 |
| EC Number | 219-610-5 |
| SMILES | CC(=O)C1=CC(=C(C(=C1)OC)O)OC |
| Density | 1.2$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 117 - 121 $degree$C (Expl.) |
| Boiling point | 334.7$+/-$37.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 131.7$+/-$20.0 $degree$C (Calc.)* |
| Index of refraction | 1.528 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Acetosyringone is a naturally occurring phenolic compound belonging to the class of methoxy-substituted aromatic ketones. Its chemical structure consists of a syringyl aromatic ring bearing two methoxy groups and one hydroxyl group, together with an acetyl substituent attached directly to the benzene ring. The compound is also known chemically as 4-hydroxy-3,5-dimethoxyacetophenone. The molecular framework of acetosyringone is derived from acetophenone, with additional methoxy substituents at the 3- and 5-positions and a hydroxyl group at the 4-position of the aromatic ring. This substitution pattern gives rise to a highly conjugated aromatic system with both electron-donating methoxy groups and a phenolic hydroxyl group. These functionalities strongly influence the compound’s chemical reactivity and biological interactions. Acetosyringone occurs naturally in various plant species and is formed as part of plant secondary metabolism. It is particularly associated with lignin-related phenolic biosynthetic pathways derived from phenylpropanoid metabolism. Syringyl-type aromatic compounds such as acetosyringone are structurally related to lignin monomers found in hardwood plants. One of the most significant scientific applications of acetosyringone arises from its role in plant–microbe interactions. It is well known as a signaling molecule involved in activation of virulence (vir) genes in Agrobacterium tumefaciens. When plants are wounded, phenolic compounds including acetosyringone can be released from damaged tissues. Agrobacterium detects these molecules through a regulatory signaling system, which activates genes required for transfer of T-DNA into plant cells. Because of this biological role, acetosyringone became highly important in plant biotechnology and genetic engineering. It is routinely added to Agrobacterium-mediated transformation systems to enhance vir gene induction and improve efficiency of DNA transfer into plant tissues. This application has made acetosyringone a standard reagent in many plant molecular biology protocols. Chemically, acetosyringone exhibits reactivity characteristic of phenolic aromatic ketones. The phenolic hydroxyl group can undergo oxidation, esterification, and etherification reactions, while the acetyl group participates in nucleophilic addition and condensation chemistry. The methoxy substituents donate electron density into the aromatic ring, influencing electrophilic substitution patterns and redox behavior. The compound can be synthesized through acetylation and methoxylation reactions starting from appropriately substituted phenolic precursors. Laboratory synthesis often involves Friedel–Crafts acylation or oxidation of corresponding alcohol intermediates. It may also be isolated from plant-derived materials. From a physicochemical standpoint, acetosyringone is moderately polar due to the presence of hydroxyl, methoxy, and ketone functionalities. The aromatic ring contributes hydrophobic character, while the oxygen-containing groups allow hydrogen bonding and dipolar interactions. These properties influence its solubility and chromatographic behavior. The conjugated aromatic ketone structure also contributes to ultraviolet absorption characteristics, which are useful in analytical detection methods. Acetosyringone can undergo oxidation under certain conditions because phenolic compounds are susceptible to redox transformations. Historically, the discovery of acetosyringone’s role in Agrobacterium signaling significantly advanced understanding of plant genetic transformation. Identification of phenolic vir gene inducers helped establish the molecular basis of Agrobacterium-mediated DNA transfer, which later became a major tool in plant biotechnology and transgenic crop development. Overall, acetosyringone is a naturally occurring methoxy-substituted phenolic ketone with important biological and biotechnological significance. Its function as an inducer of Agrobacterium virulence genes has made it an essential compound in plant genetic engineering, while its aromatic phenolic structure places it within the broader family of lignin-related plant secondary metabolites. References 2026. Phytophthora sojae effector Avr1d, Avh32, and Avh64 interacted with soybean U-box type E3 ubiquitin ligase GmPUB13 at distinct region as “effectors squads”. Phytopathology Research. DOI: 10.1186/s42483-026-00419-8 2026. Seizing the key nodes in glabridin biosynthesis network: identification of enzymes for producing licorice hairy roots with high levels of glabridin. Molecular Horticulture. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12961851 |
| Market Analysis Reports |