Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Chengdu Climb Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cdclimb.com | |||
 | +86 (28) 8760-8726 +86 13540107218 | |||
 | +86 (28) 8760-8725 | |||
 | sales@cdclimb.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Cyanochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cyanochem.com | |||
 | +86 (571) 8522-0831 +86 17788583750 | |||
 | +86 (571) 8522-0831 | |||
 | info@cyanochem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Nivon Specialties | India | |||
|---|---|---|---|---|
 | www.navoneindia.com | |||
 | +91 9323789882 | |||
 | nivonpharma@yahoo.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Punjab Chemicals & Crop Protection Limited | India | |||
|---|---|---|---|---|
 | www.punjabchemicals.com | |||
 | +91 (22) 2674-7900 | |||
 | +91 (22) 2673-6193 / 2673-6178 | |||
 | pharmainfo@punjabchemicals.com | |||
| Chemical manufacturer since 1975 | ||||
| Classification | Organic raw materials >> Amino compound |
|---|---|
| Name | 2-Chloro-1,3-bis(dimethylamino)trimethinium hexafluorophosphate |
| Synonyms | [(Z)-2-chloro-3-(dimethylamino)prop-2-enylidene]-dimethylazanium;hexafluorophosphate |
| Molecular Structure | 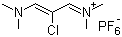 |
| Molecular Formula | C7H14ClN2.PF6 |
| Molecular Weight | 306.62 |
| CAS Registry Number | 249561-98-6 |
| EC Number | 619-785-1 |
| SMILES | CN(C)/C=C(/C=[N+](C)C)Cl.F[P-](F)(F)(F)(F)F |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H317-H319 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P272-P280-P301+P317-P302+P352-P305+P351+P338-P321-P330-P333+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
2-Chloro-1,3-bis(dimethylamino)trimethinium hexafluorophosphate is a synthetic organic salt that functions as a powerful electrophilic reagent, particularly known for its use as a dehydrating and coupling agent in organic synthesis. The compound consists of a reactive trimethinium core bearing two electron-donating dimethylamino groups and one chlorine atom, paired with a hexafluorophosphate (PF6−) counterion that provides stability and solubility in polar organic solvents. Its molecular formula is C7H17ClF6N3P. This reagent was developed to promote transformations where strong electrophilic activation is required. It is structurally related to other carbocationic reagents like the Vilsmeier reagent, but with improved reactivity due to the stabilization offered by the electron-donating amino groups and the strongly delocalized positive charge across the conjugated system. The presence of the PF6− anion enhances the reagent’s stability and reduces its hygroscopicity compared to analogous chlorides or halides. One of the major applications of 2-chloro-1,3-bis(dimethylamino)trimethinium hexafluorophosphate is in peptide synthesis and related amide bond formations. It can act as a coupling reagent that facilitates the formation of peptide bonds between carboxylic acids and amines without the need for pre-activation of the carboxylic acid. It has also been used in the synthesis of heterocycles and in the preparation of reactive intermediates where mild but powerful electrophilic conditions are needed. Its utility extends to reactions involving the activation of alcohols, carboxylic acids, and other nucleophiles. For instance, it can be employed to convert alcohols into more reactive species, facilitating substitution or elimination reactions. In carbocation chemistry, the trimethinium core serves as a stable platform for generating reactive intermediates used in rearrangements or other electrophilic processes. The compound is typically supplied as a crystalline solid, stable under dry conditions but reactive to moisture due to the presence of the electrophilic chlorine. It must be handled under inert atmosphere conditions such as in a glovebox or using Schlenk techniques. The hexafluorophosphate counterion contributes to its crystalline nature and low nucleophilicity, which helps to avoid unwanted side reactions during synthesis. Although primarily a reagent for chemical synthesis, its reactivity profile and structure have also made it a subject of study in physical organic chemistry. The delocalization of the positive charge and the resonance stabilization of the trimethinium ring are of interest in the context of carbocation stability and reactivity. In summary, 2-chloro-1,3-bis(dimethylamino)trimethinium hexafluorophosphate is a potent electrophilic reagent used in peptide coupling and other transformations requiring strong activation of nucleophilic partners. Its stability, solubility, and reactivity make it a valuable tool in synthetic organic chemistry, particularly in advanced laboratory and research settings. References 2008. Vilsmeier Reactions. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-032-00200 2005. From Vinylogous Amidinium Salts, Ketones, and Hydroxylamine. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-015-00374 2004. Etoricoxib. Pharmaceutical Substances. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-05-0110 |
| Market Analysis Reports |