Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tikslioji Sinteze | Lithuania | |||
|---|---|---|---|---|
 | www.finesynthesis.lt | |||
 | +370 (5) 213-5553 | |||
 | ardan@micro.lt | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Dafeng Chemical Com.,Ltd. | China | |||
|---|---|---|---|---|
 | www.qzdafeng.cn | |||
 | +86 16773308882 | |||
 | 927608786@qq.com | |||
 | QQ Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Paragos e. K. | Germany | |||
|---|---|---|---|---|
 | www.paragos.com | |||
 | +49 (2330) 807-9751 | |||
 | +49 (2330) 807-9752 | |||
 | sales@paragos.de | |||
| Chemical manufacturer since 2001 | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Aryl compounds >> Biphenyl compounds |
|---|---|
| Name | 4-Bromomethylbiphenyl |
| Synonyms | 1-(bromomethyl)-4-phenylbenzene |
| Molecular Structure | 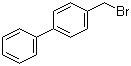 |
| Molecular Formula | C13H11Br |
| Molecular Weight | 247.13 |
| CAS Registry Number | 2567-29-5 |
| EC Number | 663-894-7 |
| SMILES | C1=CC=C(C=C1)C2=CC=C(C=C2)CBr |
| Melting point | 85 °C |
|---|---|
| Boiling point | 140 °C (10 mmHg) |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H319-H413 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P273-P280-P301+P317-P305+P351+P338-P330-P337+P317-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
4-Bromomethylbiphenyl is a significant chemical compound in the realm of organic chemistry, particularly known for its applications in synthetic chemistry and materials science. This compound features a biphenyl backbone with a bromomethyl group attached to the 4-position of one of the phenyl rings. Its unique structure has made it valuable in various chemical processes and applications. The discovery of 4-Bromomethylbiphenyl can be traced back to the development of functionalized biphenyl derivatives in the late 20th century. The compound was synthesized as part of ongoing research into the modification of biphenyl structures to create new chemical intermediates and reagents. The introduction of the bromomethyl group was intended to provide a reactive site that could be used in further chemical transformations, making it a useful tool for researchers in organic synthesis. The synthesis of 4-Bromomethylbiphenyl typically involves the bromination of biphenyl derivatives. The process begins with the preparation of biphenyl, which is commonly achieved through coupling reactions of phenyl-containing precursors. The bromomethyl group is introduced via a reaction with a brominating agent and a methylating reagent. This reaction is carried out under controlled conditions to ensure the selective addition of the bromomethyl group at the 4-position of the biphenyl ring. The resulting product is purified using techniques such as column chromatography to achieve a high-purity compound suitable for various applications. One of the primary applications of 4-Bromomethylbiphenyl is as a chemical intermediate in the synthesis of complex organic molecules. The bromomethyl group serves as a reactive handle that can participate in a variety of chemical reactions, including nucleophilic substitutions and cross-coupling reactions. This makes 4-Bromomethylbiphenyl a valuable reagent for the creation of new chemical compounds and materials. In organic synthesis, 4-Bromomethylbiphenyl is often used as a building block for the preparation of more complex biphenyl derivatives and other functionalized organic molecules. The bromomethyl group allows for the introduction of additional functional groups through subsequent chemical reactions, facilitating the development of novel compounds with specific properties. The compound is also utilized in materials science for the synthesis of advanced materials with specific electronic or optical properties. For example, 4-Bromomethylbiphenyl can be used in the preparation of organic semiconductors and polymers. The presence of the bromomethyl group can influence the electronic properties of the material, making it suitable for applications in organic electronics and optoelectronics. In addition to its use in organic synthesis and materials science, 4-Bromomethylbiphenyl can serve as a precursor for the development of pharmaceuticals and agrochemicals. The ability to modify the bromomethyl group allows for the creation of compounds with potential biological activity, expanding the scope of its applications in drug discovery and development. The advantages of using 4-Bromomethylbiphenyl include its versatility as a chemical intermediate and its ability to participate in a wide range of chemical reactions. However, challenges may include the need for careful handling of brominating agents and optimization of reaction conditions to achieve the desired product. Future research involving 4-Bromomethylbiphenyl may focus on exploring new applications and optimizing its use in various chemical processes. Researchers may also investigate modifications to the compound's structure to enhance its reactivity and develop new derivatives with improved properties. References 2020. 2-Amino- and 2-hydroxymethylbenzimidazolium bromides as protein tyrosine phosphatase 1В (PTP1В) inhibitors and other targets associated with diabetes mellitus. Russian Chemical Bulletin, 69(4). DOI: 10.1007/s11172-020-2832-5 2013. Synthesis and characterization of a series of chiral NHC-Pd complexes derived from l-phenylalanine. Transition Metal Chemistry, 38(3). DOI: 10.1007/s11243-013-9694-8 2005. Photochemical reactions of triplet p-phenylbenzyl derivatives studied by using laser flash triplet-sensitization techniques. Photochemical & photobiological sciences, 4(3). DOI: 10.1039/b416457a |
| Market Analysis Reports |