Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Jiangsu Mengde New Materials Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chinamengde.com | |||
 | +86 (511) 8686-2404 +86 18021229333 | |||
 | +86 (511) 8686-9404 | |||
 | oliverhang@chinamengde.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 1996 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Silworld Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.silworldchemical.com | |||
 | +86 (27) 8561-3400 +86 15827173649 | |||
 | +86 (27) 8561-3419 | |||
 | info@silworldchemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Alcatraz Chemicals | India | |||
|---|---|---|---|---|
 | www.alcatrazchemicals.com | |||
 | +91 (265) 234-0422 | |||
 | +91 (265) 235-3920 | |||
 | shine@alcatrazchemicals.com | |||
| Chemical manufacturer since 1985 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Wuhan Pinuo Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pinuochem.com | |||
 | +86 18427058543 | |||
 | sale01@pinuochem.com | |||
 | QQ Chat | |||
 | WeChat: Wuhan Pinuo Chemical Co.,Ltd. | |||
 | WhatsApp:Wuhan Pinuo Chemical Co.,Ltd. | |||
| Chemical distributor since 2021 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Chemical reagent >> Organic reagent >> Imide |
|---|---|
| Name | Dibenzenesulfonimide |
| Synonyms | N-(Phenylsulphonyl)benzenesulphonamide; Bis(benzene sulphonyl)-imide; BBI |
| Molecular Structure | 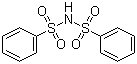 |
| Molecular Formula | C12H11NO4S2 |
| Molecular Weight | 297.34 |
| CAS Registry Number | 2618-96-4 |
| EC Number | 220-051-4 |
| SMILES | C1=CC=C(C=C1)S(=O)(=O)NS(=O)(=O)C2=CC=CC=C2 |
| Melting point | 150-155 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Dibenzenesulfonimide is an organosulfur compound that contains two benzenesulfonyl groups attached to a nitrogen atom. Its discovery came about as part of efforts to develop sulfur-containing compounds that could serve as versatile reagents in organic synthesis and materials science. The structure of dibenzenesulfonimide allows it to act as both a strong acid and a reactive intermediate, making it valuable in a variety of chemical processes. The synthesis of dibenzenesulfonimide involves the reaction of benzenesulfonyl chloride with ammonia or an amine, followed by further reaction to introduce a second benzenesulfonyl group. This process yields a stable compound with a highly electrophilic nitrogen atom that can participate in nucleophilic substitution reactions. Its ability to donate and accept electrons makes it suitable for catalyzing reactions or acting as an intermediate in the formation of more complex molecules. Dibenzenesulfonimide has found significant application in the development of pharmaceuticals and other bioactive compounds. In medicinal chemistry, it is used as a reagent for the introduction of sulfonamide groups into drug candidates. Sulfonamides are an important class of compounds known for their antibacterial properties, and the use of dibenzenesulfonimide in synthesis has facilitated the creation of new sulfonamide-containing drugs with improved efficacy and stability. In organic synthesis, dibenzenesulfonimide is employed as a reagent for electrophilic aromatic substitution reactions. It can introduce sulfonyl groups into aromatic rings, which is useful for modifying the chemical properties of organic compounds. This sulfonation process is essential in the preparation of dyes, pigments, and other specialty chemicals. The compound's ability to act as a sulfonating agent has made it an important tool for chemists working on the development of advanced materials and intermediates for industrial applications. Dibenzenesulfonimide has also been explored in polymer chemistry, where it is used to introduce sulfonamide groups into polymer structures. This modification enhances the thermal and chemical stability of polymers, making them suitable for use in harsh environments or applications requiring durable materials. The compound's ability to enhance the properties of polymers has led to its incorporation into high-performance coatings, adhesives, and other industrial products. References 2014. N-Fluorobenzenesulfonimide: An Efficient Nitrogen Source for C-N Bond Formation. Synthesis. DOI: 10.1055/s-0034-1379396 2021. N-Selenocyanato-Dibenzenesulfonimide: A New Electrophilic Selenocyanation Reagent. Synthesis. DOI: 10.1055/a-1493-6885 2015. Synthesis of Ketenaminals via Catalyst-Free Hydroamination of Ynamides and Diphenylsulfonimide. Synthesis. DOI: 10.1055/s-0034-1379937 |
| Market Analysis Reports |