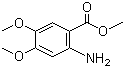
Methyl 2-amino-4,5-dimethoxybenzoate is an organic compound that belongs to the class of aromatic esters and is derived from benzoic acid. This compound features a benzene ring substituted with methoxy groups at the 4 and 5 positions, as well as an amino group at the 2-position of the benzene ring, with a methyl ester group attached to the carboxyl group. The combination of functional groups in this compound gives it interesting chemical and biological properties.
The discovery and application of methyl 2-amino-4,5-dimethoxybenzoate have been mainly driven by its potential uses in medicinal and pharmaceutical chemistry. The functional groups present in the molecule, such as the amino and methoxy groups, can influence the compound's biological activity, making it useful in the design of drugs or other bioactive molecules.
The methoxy groups are electron-donating, which can enhance the stability of the aromatic ring and affect the compound’s reactivity and interactions with other molecules. The amino group is a common functional group in pharmaceutical chemistry and can serve as a site for further chemical modifications or interactions with biological targets. This structural configuration makes methyl 2-amino-4,5-dimethoxybenzoate an interesting compound for use in drug development.
One of the most important potential applications of methyl 2-amino-4,5-dimethoxybenzoate is as a precursor or intermediate in the synthesis of biologically active molecules. The amino and methoxy groups provide sites for further modifications, allowing synthetic chemists to design more complex compounds with potential pharmaceutical activity. For example, derivatives of this compound may have applications in the treatment of diseases that involve neurological, cardiovascular, or inflammatory pathways, where the properties of methoxy and amino groups can influence molecular interactions with specific targets.
In addition to its use in pharmaceutical research, methyl 2-amino-4,5-dimethoxybenzoate may also have applications in the development of agrochemicals, particularly herbicides or fungicides. The presence of the methoxy and amino groups could influence the compound’s ability to interact with plant enzymes or other biological systems, making it a candidate for further testing in crop protection chemistry.
The compound may also have applications in the field of material science, especially in the development of organic electronic materials or as a building block for the design of polymers or other materials. The structure of methyl 2-amino-4,5-dimethoxybenzoate, with its aromatic ring and functional groups, could provide properties useful for optoelectronic devices or as an intermediate in the synthesis of complex organic materials.
Furthermore, compounds with similar structures to methyl 2-amino-4,5-dimethoxybenzoate have been explored in the context of their antioxidant and anti-inflammatory activities. The presence of electron-donating groups like the methoxy groups can enhance the bioavailability and stability of such compounds, potentially increasing their effectiveness in medical applications, especially in treating oxidative stress-related diseases.
In summary, methyl 2-amino-4,5-dimethoxybenzoate is a versatile compound with potential applications in pharmaceuticals, agrochemicals, and materials science. Its functional groups, including the methoxy and amino groups, make it a valuable intermediate in the synthesis of bioactive molecules. As research into its properties and derivatives continues, methyl 2-amino-4,5-dimethoxybenzoate could play an important role in advancing drug discovery, agrochemical development, and materials innovation.
|




















 GHS07 Warning Details
GHS07 Warning Details