Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Angewchem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.angewchem.com | |||
 | +86 18917157435 | |||
 | dylan@angewchem.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Xi'an Yinherb Bio-tech Co., Ltd | China | |||
|---|---|---|---|---|
 | yinherb.ecer.com | |||
 | +86 (29) 6333-2330 | |||
 | sales@yinherb-nutraceuticals.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Dalton Pharma Services | Canada | |||
|---|---|---|---|---|
 | www.dalton.com | |||
 | +1 (416) 661-2102 | |||
 | +1 (416) 661-2108 | |||
 | chemist@dalton.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antipyretic analgesics |
|---|---|
| Name | Paradol |
| Synonyms | [6]-Gingerone; [6]-Paradol; Heptyl 4-hydroxy-3-methoxyphenethyl ketone |
| Molecular Structure | 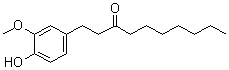 |
| Molecular Formula | C17H26O3 |
| Molecular Weight | 278.39 |
| Protein Sequence | v |
| CAS Registry Number | 27113-22-0 |
| EC Number | 248-228-1 |
| SMILES | CCCCCCCC(=O)CCC1=CC(=C(C=C1)O)OC |
| Solubility | water: practically insoluble (0.05 g/L) (25 °C), Calc.*, 10 mg/mL (Expl.), DMSO: >80 mg/mL, Ethanol: >80 mg/mL (Expl.) |
|---|---|
| Density | 1.016±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 31 - 32 °C (Expl.) |
| Boiling point | 406.4±30.0 °C 760 mmHg (Calc.)* |
| Flash point | 140.0±18.1 °C (Calc.)* |
| Index of refraction | 1.507 (Calc.)*, 1.5232 (589.3 nm 20 °C) (Expl.)** |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2014 ACD/Labs) |
| ** | Nelson, E. K. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H317 Details | ||||||||
| Safety Statements | P261-P272-P280-P302+P352-P321-P333+P317-P362+P364-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
Paradol is a naturally occurring vanilloid compound chemically known as 1‑(4‑hydroxy‑3‑methoxyphenyl)decan‑3‑one (commonly referred to as 6‑Paradol). It is found in grains of paradise (Aframomum melegueta) and ginger (Zingiber officinale). As a biotransformation product of 6‑shogaol, it retains a vanillyl functional group and exhibits a range of biological activities without the pungency associated with shogaol. Paradol’s discovery stems from studies of ginger-based phytochemicals and their metabolites. When ginger is processed or subject to microbial reduction, the highly pungent 6‑shogaol is converted into the non-pungent paradols. Among these, 6‑Paradol has emerged as a principal bioactive metabolite. Early characterization identified it as a milder, more stable metabolite useful for biological studies. It is now recognized as the main active principle in grains of paradise and ginger, contributing to the flavor but also the functional bioactivity of these botanicals ([ScienceDirect][1], [FooDB][2]). Research has demonstrated that 6‑Paradol exhibits anti‑obesity effects in preclinical models. In high-fat diet–induced obese mice, oral administration of 6‑Paradol reduced weight gain and visceral fat accumulation over two weeks, even in the absence of AMPK activation, unlike structurally related vanilloids such as 6‑gingerol and 6‑shogaol ([PubMed][3]). Neuroprotective properties were reported in rodent models of cerebral ischemia. In mice subjected to middle cerebral artery occlusion, 6‑Paradol administered after reperfusion reduced brain infarct size and improved neurological outcomes. Mechanistically, it attenuated microglial activation, inducible nitric oxide synthase (iNOS), and TNF‑α levels, suggesting potent anti‑inflammatory effects in the central nervous system ([PLOS][4]). Additional research has revealed that 6‑Paradol possesses anticancer activity. In pancreatic cancer models, it suppressed proliferation and metastasis by promoting proteasomal degradation of epidermal growth factor receptor (EGFR) and inhibiting PI3K/AKT signaling, leading to reduced invasion and migration in vitro and in vivo ([BioMed Central][5]). Other studies have shown antioxidant and anti‑inflammatory functions both in vitro and in vivo, supporting its role as a bioactive metabolite with therapeutic potential. Its non-pungent nature makes it more tolerable for long-term use compared to related ginger components ([PMC][6], [biomolther.org][7]). Potential applications of 6‑Paradol include its use as a dietary supplement component for metabolic health, inclusion in formulations targeting neuroinflammation or ischemic injury, and exploration as an anticancer agent in tumors sensitive to EGFR/AKT modulation. Its combination of safety, bioactivity, and natural origin positions it as a promising candidate for nutraceutical or therapeutic development. Limitations include the need for human clinical data and understanding of pharmacokinetics and long‑term safety. Nonetheless, preclinical findings consistently support its efficacy across multiple systems, including metabolic, inflammatory, neurological, and oncological. In summary, 6‑Paradol is a bioactive vanilloid discovered as a metabolite of 6‑shogaol and found in ginger and grains of paradise. It shows anti‑obesity, neuroprotective, anticancer, antioxidant, and anti‑inflammatory activities in preclinical studies. Its lack of pungency and healthy safety profile make it a subject of growing interest for further research and development. References Hattori H, Mori T, Shibata T, Kita M, Mitsunaga T (2021) 6‑Paradol acts as a potential anti‑obesity vanilloid from grains of paradise. Molecular Nutrition and Food Research 65 16 2100185 DOI: 10.1002/mnfr.202100185 Gaire BP, Kwon OW, Park SH, Chun K‑H, Kim SY, Shin DY et al. (2015) Neuroprotective effect of 6‑Paradol in focal cerebral ischemia involves the attenuation of neuroinflammatory responses in activated microglia. PLoS ONE 10 3 e0120203 DOI: 10.1371/journal.pone.0120203 Chung WY, Jung YJ, Surh YJ, Lee SS, Park KK (2001) Antioxidative and antitumor promoting effects of [6]‑Paradol and its homologs. Mutation Research 496 1‑2 199–206 DOI: 10.1016/S1383-5718(01)00221-2 |
| Market Analysis Reports |