Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hengshui Haoye Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemcoms.com | |||
 | +86 (318) 210-2300 +86 18632882519 | |||
 | +86 (318) 210-3390 | |||
 | firmcn@gmail.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hebei Brant Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.brantpharm.com | |||
 | +86 18713118368 | |||
 | lisa@brantpharm.com | |||
 | WhatsApp:0086 18713118368 | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Jinzhou Zhongshi Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jzzhongshi.com | |||
 | +86 (311) 8448-7556 | |||
 | +86 (311) 8448-7368 | |||
 | zhongshichemical@163.com | |||
| Chemical manufacturer since 2005 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound |
|---|---|
| Name | 4-Pyridylmercapto acetyl chloride hydrochloride |
| Synonyms | 2-pyridin-4-ylsulfanylacetyl chloride;hydrochloride |
| Molecular Structure | 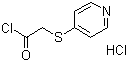 |
| Molecular Formula | C7H6ClNOS.HCl;C7H7Cl2NOS |
| Molecular Weight | 224.11 |
| CAS Registry Number | 27230-51-9 |
| EC Number | 248-348-4 |
| SMILES | C1=CN=CC=C1SCC(=O)Cl.Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H312+H332-H302-H312-H332 Details | ||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P317-P302+P352-P304+P340-P317-P321-P330-P362+P364-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
|
4-Pyridylmercapto acetyl chloride hydrochloride is a functionalized pyridine derivative that combines a 4-pyridyl group with a thiol-derived acyl chloride, forming a reactive intermediate useful in organic synthesis and medicinal chemistry. Compounds of this type are designed to introduce both a nucleophilic sulfur atom and an electrophilic acyl chloride, allowing for the formation of thioesters, amides, and other functionalized derivatives. The hydrochloride form stabilizes the compound by protonating the pyridine nitrogen, improving handling and solubility in polar solvents. Structurally, the molecule contains a pyridine ring substituted at the 4-position with a mercaptoacetyl group, in which the sulfur atom is attached to a carbonyl chloride. The 4-pyridyl moiety contributes aromaticity, nitrogen basicity, and potential hydrogen-bonding interactions, which can influence binding in biological or catalytic applications. The thiol-derived acyl chloride is highly reactive, enabling nucleophilic substitution with alcohols, amines, or thiols to form thioesters, amides, or disulfides. This dual functionality makes the compound a versatile intermediate in chemical synthesis. The preparation of 4-pyridylmercapto acetyl chloride hydrochloride typically involves first converting 4-mercaptopyridine derivatives into the corresponding thioacetate, followed by selective acylation to introduce the carbonyl chloride functionality. The hydrochloride form is generated by treatment with hydrogen chloride in an appropriate solvent, which protonates the pyridine nitrogen and stabilizes the compound. These synthetic steps allow precise control over reactivity while producing a stable, isolable intermediate suitable for further transformations. This compound finds extensive use in medicinal chemistry and chemical synthesis. In drug discovery, it can be employed as a reactive intermediate for the preparation of bioactive thioesters, amides, and heterocyclic derivatives. The presence of the pyridine nitrogen allows for coordination with metal ions in metalloenzymes or catalysts, while the acyl chloride functionality enables the formation of covalent linkages with nucleophilic groups in target molecules. Such derivatives have been explored as enzyme inhibitors, metal-chelating agents, or intermediates in the synthesis of more complex pharmacophores. In addition to its pharmaceutical applications, 4-pyridylmercapto acetyl chloride hydrochloride is valuable in organic synthesis. The combination of an activated carbonyl chloride and a nucleophilic sulfur atom permits selective reactions with a variety of nucleophiles, allowing for the construction of thioesters, heterocycles, and functionalized intermediates. The pyridyl ring also serves as a site for further functionalization via electrophilic or nucleophilic aromatic substitution, expanding the chemical diversity accessible from this compound. The hydrochloride salt form improves the solubility of the compound in polar media and enhances stability during storage and handling, which is important when working with reactive acyl chlorides. This property allows chemists to perform reactions under controlled conditions and isolate high-purity products for downstream applications in synthesis or biological evaluation. Overall, 4-pyridylmercapto acetyl chloride hydrochloride is a highly functionalized heterocyclic intermediate that integrates a reactive acyl chloride, a nucleophilic sulfur, and an aromatic pyridine ring. Its structural features provide versatility in organic synthesis, medicinal chemistry, and the preparation of bioactive derivatives, making it a valuable tool for the construction of thioesters, amides, and other functionalized molecules. References 2023. A kind of synthesis method of cefapirin intermediate. CN-117343003-A. URL: https://patents.google.com/patent/CN117343003A/en 2012. Method for preparing cephapirin benzathine. CN-102020665-B. URL: https://patents.google.com/patent/CN102020665B/en |
| Market Analysis Reports |