Online Database of Chemicals from Around the World
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Amine |
|---|---|
| Name | 3-Amino-4-phenoxy-5-sulfamoylbenzoic acid |
| Molecular Structure | 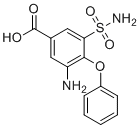 |
| Molecular Formula | C13H12N2O5S |
| Molecular Weight | 308.31 |
| CAS Registry Number | 28328-54-3 |
| EC Number | 248-971-1 |
| SMILES | C1=CC=C(C=C1)OC2=C(C=C(C=C2S(=O)(=O)N)C(=O)O)N |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
3-Amino-4-phenoxy-5-sulfamoylbenzoic acid is a chemical compound that has attracted attention in the fields of medicinal chemistry and pharmaceutical development due to its potential therapeutic applications. The compound's structure, characterized by the presence of amino, phenoxy, and sulfamoyl functional groups, provides it with a unique combination of chemical properties that are useful in various biological contexts. The discovery of 3-Amino-4-phenoxy-5-sulfamoylbenzoic acid is rooted in efforts to develop new therapeutic agents targeting specific enzymes and receptors involved in disease processes. The compound belongs to a class of benzoic acid derivatives that are often explored for their ability to interact with biological macromolecules. The presence of the sulfamoyl group is particularly significant, as it is known to mimic phosphate groups, allowing the compound to interact with proteins that bind to or process phosphorylated molecules. This makes 3-Amino-4-phenoxy-5-sulfamoylbenzoic acid a candidate for the inhibition of enzymes such as kinases or phosphatases, which are key regulators in cellular signaling pathways. One of the primary applications of this compound lies in the development of drugs for the treatment of conditions such as cancer and inflammatory diseases. The sulfamoylbenzoic acid scaffold has been studied extensively for its ability to inhibit carbonic anhydrase, an enzyme that is involved in the regulation of pH within cells and tissues. Inhibiting this enzyme can disrupt the microenvironment of tumors, making them less able to grow and metastasize. Additionally, the compound's amino group allows for further chemical modifications, enabling the design of derivatives with enhanced potency or selectivity for specific isoforms of the enzyme. In addition to its potential in cancer therapy, 3-Amino-4-phenoxy-5-sulfamoylbenzoic acid has shown promise in the treatment of bacterial infections. The compound's structure allows it to inhibit bacterial enzymes that are crucial for survival, such as dihydropteroate synthase, which is involved in folate synthesis. By targeting this enzyme, the compound can effectively block the production of folate, a vital nutrient for bacterial growth, leading to the death of the pathogen. This mode of action is similar to that of traditional sulfonamide antibiotics, but the presence of the phenoxy group in 3-Amino-4-phenoxy-5-sulfamoylbenzoic acid can potentially reduce the development of resistance by introducing a new structural motif that is less recognized by bacterial resistance mechanisms. The synthesis of 3-Amino-4-phenoxy-5-sulfamoylbenzoic acid involves the functionalization of a benzoic acid core with amino, phenoxy, and sulfamoyl groups. The synthesis is typically achieved through a series of substitution reactions, where the phenoxy group is introduced via nucleophilic aromatic substitution, followed by the introduction of the sulfamoyl group through sulfonation reactions. The final step involves the introduction of the amino group, which can be accomplished through a variety of methods, including direct amination or reductive amination of a nitro precursor. The versatility of 3-Amino-4-phenoxy-5-sulfamoylbenzoic acid in medicinal chemistry makes it a valuable scaffold for drug discovery. Its ability to interact with a wide range of biological targets, coupled with the ease of synthesis and potential for further modification, ensures that it will continue to be a compound of interest in the development of new therapeutics. As research continues, this compound may pave the way for the development of new drugs that address unmet medical needs in areas such as oncology and infectious diseases. References 2008. Effects of water deprivation on drug pharmacokinetics: Correlation between drug metabolism and hepatic CYP isozymes. Archives of Pharmacal Research, 31(8). DOI: 10.1007/s12272-001-1269-3 2003. Investigation of the photochemical properties and in vitro phototoxic potential of bumetanide. Photochemical & Photobiological Sciences, 2(10). DOI: 10.1039/b302922k 1994. Pharmacokinetics and pharmacodynamics of bumetanide after intravenous and oral administration to rats: Absorption from various GI segments. Journal of Pharmacokinetics and Biopharmaceutics, 22(1). DOI: 10.1007/bf02353407 |
| Market Analysis Reports |