Online Database of Chemicals from Around the World
| Gou Hebei Chemicals | India | |||
|---|---|---|---|---|
 | gouhebeichems.store | |||
 | +86 15874272473 | |||
 | info@gouhebeichems.store | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Classification | Analytical chemistry >> Standard >> Standard material |
|---|---|
| Name | alpha-Methyl-6-benzofuranethanamine hydrochloride |
| Synonyms | 1-(1-benzofuran-6-yl)propan-2-amine;hydrochloride |
| Molecular Structure | 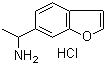 |
| Molecular Formula | C11H13NO.HCl |
| Molecular Weight | 211.69 |
| CAS Registry Number | 286834-84-2 |
| EC Number | 803-417-3 |
| SMILES | CC(CC1=CC2=C(C=C1)C=CO2)N.Cl |
| Hazard Symbols |  GHS09 Warning Details GHS09 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H411 Details | ||||||||
| Safety Statements | P273-P391-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
α-Methyl-6-benzofuranamine hydrochloride, commonly known as 6-APB, is a compound recognized for its psychoactive effects and potential therapeutic applications. 6-APB was first synthesized and studied in the late 1990s as part of the search for novel psychoactive substances. Its structure features a benzofuran ring fused to the amphetamine backbone with a methyl substitution at the α position, which distinguishes it from other hallucinogens and stimulants. 2-Amino-1-phenylethan-1-one condenses with 1-(2-benzofuranyl)propan-2-amine under controlled conditions to form 6-APB hydrochloride. Pharmacologically, 6-APB acts as a serotonin-norepinephrine-dopamine releasing agent (SNDRA), promoting the release of these neurotransmitters in the brain. Its primary mechanism of action involves binding to and promoting the release of serotonin, dopamine, and norepinephrine from presynaptic neurons. This activity can lead to elevated moods, increased empathy, and heightened sensory perceptions, characteristics that contribute to its recreational and potential therapeutic effects. Due to its euphoric stimulant properties, 6-APB is highly sought after in recreational drug use settings and is typically taken orally in tablet or capsule form. Users report feelings of euphoria, increased sociability, and heightened sensory experiences, similar to the sensations induced by MDMA. However, like other psychoactive substances, 6-APB carries risks, including adverse effects such as increased heart rate, hypertension, and potential psychological dependence. In addition to recreational use, research has explored the therapeutic potential of 6-APB for a variety of conditions. Its ability to modulate serotonin and dopamine levels suggests it has the potential to treat mood disorders, including depression and anxiety. Some studies have also investigated its use in psychotherapy settings to enhance empathy and communication, but further clinical research is needed to determine its safety and effectiveness. The regulatory status of 6-APB varies across the globe, with many countries implementing restrictions or outright bans due to its psychoactive effects and potential for abuse. In some jurisdictions, it is classified as a controlled substance, restricting its availability and use to authorized medical and research purposes. References none |
| Market Analysis Reports |