Online Database of Chemicals from Around the World
| MolScanner | Singapore | |||
|---|---|---|---|---|
 | www.molscanner.com | |||
 | +86 18621675448 | |||
 | marketing@molscanner.com | |||
 | WhatsApp:9896 7603 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Pyrroles |
|---|---|
| Name | (S,Z)-(2-(Fluoromethylene)tetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol hydrochloride |
| Synonyms | [(6Z,8S)-6-(fluoromethylidene)-2,3,5,7-tetrahydro-1H-pyrrolizin-8-yl]methanol;hydrochloride |
| Molecular Structure | 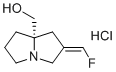 |
| Molecular Formula | C9H15ClFNO |
| Molecular Weight | 207.67 |
| CAS Registry Number | 2871770-04-4 |
| SMILES | C1C[C@]2(C/C(=C/F)/CN2C1)CO.Cl |
|
The chemical substance (S,Z)-(2-(fluoromethylene)tetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol hydrochloride is a chiral pyrrolizine derivative featuring a fluoromethylene group and a hydroxymethyl substituent in its hydrochloride salt form, valued as a synthetic intermediate in pharmaceutical chemistry. Its discovery and applications are well-documented in the literature, rooted in the development of pyrrolizine chemistry, fluorinated heterocycles, and stereoselective synthesis. The origins of this compound are linked to the study of pyrrolizines, bicyclic heterocycles with two fused five-membered rings sharing a bridgehead nitrogen, explored since the early 20th century for their presence in alkaloids and pharmacological potential. The tetrahydro-1H-pyrrolizine core, a fully saturated pyrrolizine, gained attention in the mid-20th century for its structural similarity to bioactive natural products. The introduction of a fluoromethylene group, a fluorinated exocyclic alkene, became feasible in the late 20th century with advances in fluorination and olefination techniques, driven by the pharmaceutical industry’s interest in fluorine’s ability to enhance metabolic stability and lipophilicity. The chiral (S)-hydroxymethyl group and the (Z)-configuration of the fluoromethylene emerged to meet the demand for enantiopure intermediates with specific stereochemistry, supported by progress in asymmetric synthesis during the 1970s and 1980s. The hydrochloride salt form enhances solubility and stability for synthetic applications. Synthetically, (S,Z)-(2-(fluoromethylene)tetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol hydrochloride is prepared through a multi-step process. A typical route starts with a chiral pyrrolidine precursor, such as (S)-proline, which is cyclized with a suitable carbonyl compound to form the tetrahydro-1H-pyrrolizine core. The hydroxymethyl group at the 7a-position is introduced via stereoselective addition or reduction of a carbonyl precursor, using a chiral catalyst or auxiliary to ensure the (S)-configuration. The 2-(fluoromethylene) group is installed through a Wittig-type reaction or fluorinated Horner-Wadsworth-Emmons olefination, reacting a 2-keto-pyrrolizine with a fluorinated phosphonate reagent to achieve the (Z)-configuration. The resulting free base is converted to the hydrochloride salt by treatment with hydrochloric acid to improve crystallinity and handling. These steps rely on well-established protocols in heterocyclic synthesis, chiral chemistry, and fluorination, ensuring high stereochemical and geometric purity. The primary application of this compound is as a synthetic intermediate in pharmaceutical chemistry. The pyrrolizine core is a privileged scaffold in alkaloid-inspired drugs targeting neurological disorders, cancer, and pain management, due to its compact, rigid structure. The (S)-hydroxymethyl group provides a chiral, polar handle for esterification, etherification, or oxidation, while the (Z)-fluoromethylene group enhances metabolic stability and offers a reactive site for cycloadditions or conjugate additions. The hydrochloride salt form improves solubility, facilitating reactions in aqueous systems. This compound is frequently used in the synthesis of drug candidates, such as opioid receptor modulators, kinase inhibitors, and antitumor agents, where the fluorinated pyrrolizine optimizes binding affinity and pharmacokinetic properties. In academic research, the compound is employed to study stereoselective synthesis, fluoromethylene reactivity, and pyrrolizine functionalization. Its synthesis has contributed to advancements in fluorinated olefination and chiral heterocyclic chemistry. The compound also finds use in synthesizing specialty chemicals, such as fluorinated probes or ligands, where the chiral and fluorinated features are advantageous. The significance of (S,Z)-(2-(fluoromethylene)tetrahydro-1H-pyrrolizin-7a(5H)-yl)methanol hydrochloride lies in its role as a chiral, fluorinated intermediate combining the biological relevance of pyrrolizines with the synthetic versatility of fluoromethylene and hydroxymethyl groups. Its development reflects progress in stereoselective synthesis, fluorination, and heterocyclic chemistry. By enabling the efficient synthesis of enantiopure, biologically active molecules, it has become a critical tool in advancing pharmaceutical and chemical research. |
| Market Analysis Reports |