Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Mascot I.E. CO.,Ltd. | China | |||
|---|---|---|---|---|
 | www.mascotchem.com | |||
 | +86 (519) 8501-0339 +86 13584504415 | |||
 | +86 (519) 8501-0366 | |||
 | info@mascotchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2006 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Zouping Mingxing Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zoutong.com.cn | |||
 | +86 13605431940 | |||
 | +86 (543) 224-0079 | |||
 | sdzpmxchem@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Yancheng Lvye Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lvyechem.com | |||
 | +86 (515) 8987-6101 | |||
 | +86 (515) 8987-6199 | |||
 | daiyafei@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Nanjing MSN Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.msnchem.com | |||
 | +86 (25) 5701-5632 +86 18013328036 | |||
 | +86 (25) 5701-9235 | |||
 | info@msnchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Silworld Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.silworldchemical.com | |||
 | +86 (27) 8561-3400 +86 15827173649 | |||
 | +86 (27) 8561-3419 | |||
 | info@silworldchemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Glentham Life Sciences | UK | |||
|---|---|---|---|---|
 | www.glenthamls.com | |||
 | +44 (2033) 978-798 | |||
 | +44 (2033) 978-909 | |||
 | info@glenthamls.com | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Linyi Lab Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.labspharm.com | |||
 | +86 (539) 782-9635 | |||
 | +86 (539) 782-9635 | |||
 | Alex001@labspharm.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shandongjunrui Pharmaceutials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yanchem.com | |||
 | +86 (539) 563-6807 | |||
 | 2676735117@qq.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Hubei guoheng pharmaceutical technology co., Ltd | China | |||
|---|---|---|---|---|
 | www.ghpharmatech.com | |||
 | +86 15572577573 | |||
 | 1332578202@qq.com | |||
| Chemical distributor since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Richman Chemical Inc. | USA | |||
|---|---|---|---|---|
 | www.richmanchemical.com | |||
 | +1 (215) 628-2946 | |||
 | +1 (215) 628-4262 | |||
 | kac@richmanchemical.com | |||
| Chemical manufacturer since 1988 | ||||
| chemBlink Standard supplier since 2022 | ||||
| J&H Chemical HK Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jhechem.com | |||
 | +86 (571) 8739-6430 8739-6432 | |||
 | +86 (571) 8739-6431 | |||
 | sales@jhechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Shanghai Junrao Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.junrao.com | |||
 | +86 (21) 3225-0215 | |||
 | +86 (21) 6257-6357 | |||
 | sales@junrao.com | |||
| Chemical manufacturer since 2005 | ||||
| City Chemical LLC | USA | |||
|---|---|---|---|---|
 | www.citychemical.com | |||
 | +1 (203) 932-2489 | |||
 | +1 (203) 937-8400 | |||
 | sales@citychemical.com | |||
| Chemical distributor | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| IOLITEC Ionic Liquids Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.iolitec-usa.com | |||
 | +1 (205) 348-2831 | |||
 | +1 (205) 348-3510 | |||
 | info@iolitec-usa.com | |||
| Chemical manufacturer since 2002 | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Cambridge Isotope Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.isotope.com | |||
 | +1 (978) 749-8000 | |||
 | +1 (978) 749-2768 | |||
 | cilsales@isotope.com | |||
| Chemical manufacturer | ||||
| Alfa Aesar. | USA | |||
|---|---|---|---|---|
 | www.alfa.com | |||
 | +1 (978) 521-6300 | |||
 | +1 (978) 521-6350 | |||
 | info@alfa.com | |||
| Chemical manufacturer | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Shanghai 3W Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.3wchem.com | |||
 | +86 (21) 6380-4615 5355-0305 | |||
 | +86 (21) 6381-4984 / 5371-2862 | |||
 | sale@3wchem.com | |||
| Chemical manufacturer since 1993 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Classification | Chemical pesticide >> Fungicide >> Azole fungicide |
|---|---|
| Name | Imidazole |
| Synonyms | 1,3-Diazole; 1H-Imidazole; Glyoxaline |
| Molecular Structure | 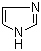 |
| Molecular Formula | C3H4N2 |
| Molecular Weight | 68.08 |
| CAS Registry Number | 288-32-4 |
| EC Number | 206-019-2 |
| SMILES | C1=CN=CN1 |
| Density | 1.1±0.1 g/cm3 Calc.*, 1.03 g/mL (Expl.) |
|---|---|
| Melting point | 88 - 91 °C (Expl.) |
| Boiling point | 257.0±9.0 °C 760 mmHg (Calc.)*, 256 °C (Expl.) |
| Flash point | 145.0 °C (Calc.)*, 145 °C (Expl.) |
| Solubility | 10 mM (H2O) (Expl.) |
| Index of refraction | 1.528 (Calc.)*, 1.48 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS05;GHS07;GHS08 Danger Details GHS05;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H314:-H360D: Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P264-P270-P280-P301+P317-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P318-P321-P330-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1759 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Imidazole is a five-membered planar heteroaromatic compound composed of three carbon atoms and two nitrogen atoms at the 1 and 3 positions in the ring. Its molecular formula is C3H4N2, and it has a molar mass of approximately 68.08 g/mol. Imidazole is an important basic building block in organic and biological chemistry due to its aromatic character, amphoteric nature, and ability to participate in hydrogen bonding and metal coordination. Imidazole was first synthesized in the mid-19th century by Heinrich Debus in 1858, who discovered it during experiments involving glyoxal, ammonia, and formaldehyde. The synthesis, now known as the Debus synthesis, remains a foundational method for producing substituted imidazoles and was later refined by researchers such as James Moir and Arthur Hantzsch. The Hantzsch synthesis, which involves the condensation of 1,2-dicarbonyl compounds with aldehydes and ammonia or amines, is widely used in modern laboratories for the preparation of imidazole derivatives. Imidazole is a colorless to pale yellow crystalline solid at room temperature and has a melting point of about 89-91 °C. It is highly soluble in water and polar organic solvents, making it useful in both aqueous and non-aqueous systems. The ring system is aromatic due to the presence of six delocalized π electrons, including a lone pair from the pyrrole-like nitrogen and the double bonds within the ring. One nitrogen atom is sp2-hybridized with a lone pair contributing to aromaticity (position 1), while the other nitrogen is similar to pyridine (position 3), bearing a lone pair that does not participate in aromaticity, making it available for protonation or hydrogen bonding. Biologically, the imidazole ring is of great significance. It is a component of the amino acid histidine, which is essential for protein structure and enzyme function. In many metalloproteins and enzymes, the imidazole group of histidine acts as a ligand that coordinates metal ions such as Fe2+, Zn2+, or Cu2+, playing a critical role in catalysis, electron transfer, and structural stability. Histidine residues are also involved in acid-base catalysis due to the proton-donating and -accepting capabilities of the imidazole group. Imidazole and its derivatives are widely used in pharmaceutical chemistry. Many antifungal drugs, including clotrimazole, ketoconazole, and miconazole, contain imidazole rings. These compounds typically inhibit fungal cytochrome P450 enzymes, disrupting ergosterol biosynthesis and compromising the integrity of fungal cell membranes. Imidazole-containing compounds are also used in the treatment of cancer, hypertension, and neurological disorders, due to their bioactive properties and structural versatility. In synthetic chemistry, imidazole serves as a useful base and nucleophilic catalyst. It is commonly used in peptide coupling reactions, nucleoside chemistry, and the activation of carboxylic acids. The ring can be substituted at various positions, allowing for the development of tailored molecules for specific chemical or biological purposes. Methylation, halogenation, and alkylation reactions are frequently employed to modify the imidazole ring. Imidazole derivatives are also employed in coordination chemistry and materials science. Due to the basic nature of the ring and its nitrogen lone pairs, imidazole readily forms complexes with transition metals. These complexes can be used in catalysis, molecular recognition, and the development of supramolecular assemblies. From an analytical perspective, imidazole is identified and studied using techniques such as NMR spectroscopy, where the ring protons and nitrogen environments produce characteristic signals. Infrared spectroscopy detects the N-H stretch and ring vibrations, while mass spectrometry and elemental analysis confirm molecular weight and composition. In conclusion, imidazole is a simple yet highly versatile heterocycle with broad applications in organic synthesis, medicinal chemistry, and biochemistry. Its aromaticity, hydrogen bonding potential, and coordination behavior make it essential in both natural and synthetic chemical systems. References 2025. Colorimetric fluorescence of the 1,10-phenantholineyl-imidazole sensor probe for the selective detection of Zn2+ and Cd2+ ions. Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 337. DOI: 10.1016/j.saa.2024.125436 2025. Formation of amino acid-based imidazole salts considerably increased the determined level of fluorescent advanced glycation end products in biscuits. Food Chemistry, 469. DOI: 10.1016/j.foodchem.2024.142227 2025. Direct and quantitative analysis of tRNA acylation using intact tRNA liquid chromatography-mass spectrometry. Nature Protocols, 20(1). DOI: 10.1038/s41596-024-01086-9 |
| Market Analysis Reports |