Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Survival Technologies Pvt Ltd | India | |||
|---|---|---|---|---|
 | www.survivaltechnologies.in | |||
 | +91 (22) 4212-8221 | |||
 | +91 (22) 4212-8226 | |||
 | info@survivaltechnologies.in | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Ring Specialty Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.ringchemicals.com | |||
 | +1 (416) 493-6870 | |||
 | info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Cn Chemunion Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cnchemunion.com | |||
 | +86 17366116869 | |||
 | david@cnchemunion.com | |||
 | QQ Chat | |||
 | WeChat: +86 15250069576 | |||
 | WhatsApp:+86 17366116869 | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Changzhou Suntton Co., Ltd. | USA | |||
|---|---|---|---|---|
 | www.suntton.com | |||
 | +1 (914) 965-2077 +86 (519) 8513-6089 | |||
 | +1 (914) 375-2093 / +86 (519) 8513-6133 | |||
 | luke.verdet@suntton.com lily.chen@suntton.com | |||
| Chemical manufacturer | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Allyl chloroformate |
| Synonyms | prop-2-enyl carbonochloridate |
| Molecular Structure | 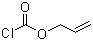 |
| Molecular Formula | C4H5ClO2 |
| Molecular Weight | 120.5 |
| CAS Registry Number | 2937-50-0 |
| EC Number | 220-916-6 |
| SMILES | C=CCOC(=O)Cl |
| Density | 1.136 |
|---|---|
| Boiling point | 74 °C (720 mmHg) |
| Refractive index | 1.422 |
| Flash point | 31 °C |
| Hazard Symbols |    GHS02;GHS05;GHS06 Danger Details GHS02;GHS05;GHS06 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H301-H314-H330 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P264-P270-P271-P280-P284-P301+P316-P301+P330+P331-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P320-P321-P330-P363-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Allyl chloroformate is an organic compound with the chemical formula C4H5ClO2. It is a member of the chloroformate family, which includes compounds known for their reactivity and versatility in organic synthesis. Allyl chloroformate is a colorless liquid with a pungent odor, and it is used primarily as a reagent in chemical reactions involving esterification and carbamoylation. The discovery of allyl chloroformate emerged from the broader investigation into chloroformates and their applications. Chloroformates are known for their utility in introducing functional groups into organic molecules, particularly in the synthesis of esters and carbamates. Allyl chloroformate was synthesized by the reaction of allyl alcohol with phosgene, a common method for preparing chloroformates. This compound's distinct reactivity is attributed to the presence of both the chloroformate and allyl groups, which enhance its utility in various chemical transformations. In organic synthesis, allyl chloroformate serves as a valuable reagent for the formation of esters and carbamates. It reacts with alcohols to produce allyl esters, which are important intermediates in the synthesis of complex organic compounds. Additionally, allyl chloroformate can be used to prepare carbamates from amines, a reaction that is useful in creating protective groups or modifying biological molecules. The allyl group in allyl chloroformate offers additional reactivity, allowing for further transformations such as cross-coupling reactions or polymerizations. The applications of allyl chloroformate extend to several industrial and research areas. In the pharmaceutical industry, it is employed in the synthesis of active pharmaceutical ingredients (APIs) and intermediates. Its ability to introduce ester and carbamate functionalities makes it valuable in the development of new drugs and therapeutic compounds. In the field of polymer chemistry, allyl chloroformate is used in the preparation of functionalized polymers with specific properties, such as increased reactivity or improved solubility. Furthermore, allyl chloroformate is utilized in the synthesis of agrochemicals and other specialty chemicals. Its reactivity allows for the creation of diverse chemical structures, contributing to the development of novel compounds with desired properties. Research into new applications and methods involving allyl chloroformate continues, highlighting its significance as a versatile reagent in organic synthesis. References 2024, Synthetic studies on the tetrasubstituted D-ring of cystobactamids lead to potent terephthalic acid antibiotics, _Communications Chemistry_, 7(1) DOI: https://doi.org/10.1038/s42004-024-01337-6 2013, Kinetic Studies that Evaluate the Solvolytic Mechanisms of Allyl and Vinyl Chloroformate Esters, _International Journal of Molecular Sciences_, 14(4) DOI: https://doi.org/10.3390/ijms14047286 2014, Synthesis of N-Allyloxycarbonyl 3,5-Dihydroxyphenylglycine via Photochemical Wolff Rearrangement-Nucleophilic Addition Sequence in a Micro-Flow Reactor, _Journal of Flow Chemistry_, 4(4) DOI: https://doi.org/10.1556/jfc-d-14-00015 |
| Market Analysis Reports |