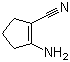
2-Amino-1-cyclopentene-1-carbonitrile is an organic compound featuring a cyclopentene ring structure substituted with an amino group and a nitrile group. Its chemical formula is C6H8N2, and it is often used in organic synthesis due to its versatility and reactivity. The compound consists of a five-membered cyclopentene ring, which is an important structural motif in organic chemistry, along with an amino group (–NH2) at the 2-position and a nitrile group (–C≡N) at the 1-position. This unique combination of functional groups makes 2-amino-1-cyclopentene-1-carbonitrile a valuable intermediate for various chemical reactions.
The discovery of 2-amino-1-cyclopentene-1-carbonitrile can be traced back to the mid-20th century, when the synthesis of substituted cyclopentene derivatives began to attract attention. The compound was initially synthesized as part of efforts to explore the reactivity and functionalization of cyclopentene rings. Over time, its potential as a versatile intermediate in organic synthesis became clear, especially in the development of more complex molecular structures.
One of the primary applications of 2-amino-1-cyclopentene-1-carbonitrile is as an intermediate in the synthesis of various organic compounds, particularly in the fields of medicinal chemistry and materials science. The presence of both the amino and nitrile groups allows the compound to participate in a variety of reactions, including nucleophilic substitution, cyclization, and condensation reactions. These reactions are used to create more complex molecules, including pharmaceutical agents, agrochemicals, and materials with specific electronic properties.
In medicinal chemistry, 2-amino-1-cyclopentene-1-carbonitrile has been investigated for its potential as a precursor to bioactive compounds. The amino group can undergo further transformations, leading to the formation of derivatives with enhanced pharmacological activity. The nitrile group can also be a key site for chemical modifications, making this compound useful in the design of novel therapeutic agents. For example, it has been explored as a building block for the synthesis of heterocyclic compounds with potential anticancer, antimicrobial, and anti-inflammatory activities.
Another area where 2-amino-1-cyclopentene-1-carbonitrile has been applied is in the development of materials with specific properties. The reactivity of the nitrile group, combined with the electron-rich nature of the cyclopentene ring, makes it a valuable building block for materials that require specific electronic or structural characteristics. This has led to its use in the synthesis of materials for electronics, sensors, and optoelectronic devices, where its ability to form stable molecular structures is crucial.
Furthermore, 2-amino-1-cyclopentene-1-carbonitrile has potential applications in the development of new catalysts. The combination of its amino and nitrile groups offers unique reactivity that can be harnessed for catalysis in organic reactions, such as carbon-carbon bond formation, which is central to the synthesis of many complex organic molecules. Its ability to coordinate with metal ions also opens the possibility for its use in metal-catalyzed reactions.
In conclusion, 2-amino-1-cyclopentene-1-carbonitrile is a highly versatile compound with a wide range of applications in organic synthesis, medicinal chemistry, materials science, and catalysis. Its unique combination of functional groups makes it an important intermediate for the creation of more complex molecules with diverse properties. Continued research into its reactivity and potential applications may lead to new discoveries and uses for this important chemical substance.
References
1995. Diethyl acetonedicarboxylate - a precursor for the synthesis of new substituted 4-aminoquinolines and fused 4-aminopyridines. Monatshefte für Chemie / Chemical Monthly, 126(3).
DOI: 10.1007/bf00816102
|



































 GHS07 Warning Details
GHS07 Warning Details