Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Classification | Biochemical >> Amino acids and their derivatives |
|---|---|
| Name | tert-butyl (2-fluoro-5-hydroxy-4-methoxyphenyl)carbamate |
| Molecular Structure | 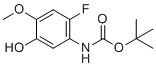 |
| Molecular Formula | C12H16FNO4 |
| Molecular Weight | 257.26 |
| CAS Registry Number | 2952660-99-8 |
| SMILES | C1(=C(C=C(C(=C1)OC)O)NC(OC(C)(C)C)=O)F |
| Density | 1.254±0.06 g/mL (20 °C, 760 Torr) |
|---|---|
| Boiling point | 308.7±42.0 °C |
|
tert-Butyl (2-fluoro-5-hydroxy-4-methoxyphenyl) carbamate is a synthetic compound used primarily in drug discovery and development. The molecule has multiple functional groups, including the tert-butyl carbamate moiety, fluorine atoms, and hydroxyl and methoxy substituents on the aromatic ring, which together account for its unique chemical behavior and potential biological activity. This compound was developed to improve the aromatic system to enhance pharmacokinetic properties. The tert-butyl carbamate group can be used as a protecting moiety for amine functional groups during organic synthesis, especially when synthesizing drug intermediates. The fluorine atom provides metabolic stability, as fluorine substitution generally prevents enzymatic degradation, thereby prolonging the activity of the compound in biological systems. The application of tert-butyl (2-fluoro-5-hydroxy-4-methoxyphenyl) carbamate is mainly seen in the synthesis of drug candidates for the treatment of various diseases. Compounds with similar structures are being investigated for their potential as kinase inhibitors, anti-inflammatory agents, and anticancer agents. The combination of fluorine and hydroxyl groups on the aromatic ring can enhance receptor binding affinity and selectivity, making this molecule a valuable scaffold for further derivatization. In synthetic chemistry, the stability of this compound makes it a key intermediate in multistep synthetic processes. The tert-butyl carbamate group can be selectively removed under mild acidic conditions, facilitating the release of the free amine for subsequent transformations. While much data on their specific applications are still emerging, this class of compounds represents a promising tool for advancing pharmaceutical research and improving drug design strategies. References none |
| Market Analysis Reports |