Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Boyuan Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.boyuanpharm.com | |||
 | +86 (531) 6995-4981 8896-3280 +86 15806417970 | |||
 | +86 (531) 8896-4879 | |||
 | Jeffrey.Liu@boyuanpharm.com boyuanchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Nanjing Bic Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bicbiotech.com | |||
 | +86 18851183712 | |||
 | sales@bicbiotech.com | |||
 | QQ Chat | |||
 | WeChat: +8618851183712 | |||
 | WhatsApp:+8618851183712 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Ryan Scientific, Inc. | USA | |||
|---|---|---|---|---|
 | www.ryansci.com | |||
 | +1 (843)-884-4911 | |||
 | +1 (843) 884-5568 | |||
 | sales@ryansci.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives |
|---|---|
| Name | 2-(tert-Butoxycarbonylamino)thiazole-5-carboxylic acid |
| Synonyms | 2-[(2-methylpropan-2-yl)oxycarbonylamino]-1,3-thiazole-5-carboxylic acid |
| Molecular Structure | 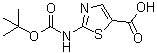 |
| Molecular Formula | C9H12N2O4S |
| Molecular Weight | 244.27 |
| CAS Registry Number | 302964-02-9 |
| EC Number | 675-956-0 |
| SMILES | CC(C)(C)OC(=O)NC1=NC=C(S1)C(=O)O |
| Solubility | Very slightly soluble (0.76 g/L) (25 $degree$C), Calc.* |
|---|---|
| Density | 1.406$+/-$0.06 g/cm3 (20 $degree$C 760 Torr), Calc.* |
| Melting point | 202 $degree$C (Decomposes) (Expl.) |
| Index of refraction | 1.602 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H317 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P272-P280-P302+P352-P321-P333+P317-P362+P364-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2-(tert-Butoxycarbonylamino)thiazole-5-carboxylic acid is a heterocyclic compound that features a thiazole ring substituted with a carboxylic acid at the 5-position and a Boc-protected amino group at the 2-position. Thiazole derivatives are important scaffolds in medicinal chemistry and organic synthesis due to their aromaticity, nitrogen and sulfur heteroatoms, and ability to participate in hydrogen bonding and π–π interactions. The Boc (tert-butoxycarbonyl) protecting group stabilizes the amino functionality during synthetic manipulations, making the compound a versatile intermediate for further functionalization. Structurally, the molecule contains a five-membered thiazole ring with the sulfur atom at position 1 and the nitrogen at position 3. The 2-position bears the Boc-protected amino group, which provides steric shielding and protects the amine from undesired reactions, while the carboxylic acid at the 5-position is available for amide bond formation, esterification, or salt formation. The combination of these functional groups makes the compound useful in the synthesis of peptides, heterocyclic analogs, and drug-like molecules. The synthesis of 2-(tert-butoxycarbonylamino)thiazole-5-carboxylic acid generally starts from thiazole-5-carboxylic acid or its esters, followed by selective amination at the 2-position and protection of the amino group using di-tert-butyl dicarbonate. This stepwise approach allows the introduction of the Boc group without affecting the carboxylic acid functionality, producing a stable intermediate suitable for downstream chemical reactions. Modern synthetic methods allow high regioselectivity and yield, ensuring reproducible preparation for research or industrial applications. In medicinal chemistry, thiazole derivatives such as this compound are widely explored as intermediates in the preparation of kinase inhibitors, enzyme modulators, and heterocyclic drug candidates. The Boc-protected amino group allows selective deprotection under acidic conditions, revealing the free amine for coupling with other molecules, including peptides or pharmacophores. The carboxylic acid provides a reactive handle for amide bond formation, which is fundamental in constructing more complex molecules with desired biological activity. Beyond pharmaceutical applications, 2-(tert-butoxycarbonylamino)thiazole-5-carboxylic acid is a versatile building block in organic synthesis. The thiazole ring can undergo electrophilic or nucleophilic substitution, cross-coupling reactions, and cyclization to form fused heterocycles. The combination of a protected amine and a free carboxylic acid enables stepwise functionalization, which is essential for preparing libraries of derivatives or functionalized intermediates in multi-step synthetic routes. The Boc group is particularly valuable because it provides chemical stability to the amino functionality while allowing controlled deprotection. This feature facilitates the synthesis of peptides, peptidomimetics, or small-molecule heterocycles where the thiazole scaffold contributes planarity, aromaticity, and potential hydrogen-bonding interactions. The carboxylic acid group can also form salts or esters, improving solubility and handling properties in synthetic protocols. Overall, 2-(tert-butoxycarbonylamino)thiazole-5-carboxylic acid exemplifies a functionalized thiazole derivative that integrates a protected amino group and a carboxylic acid on a heterocyclic scaffold. Its structural features provide chemical versatility for peptide synthesis, heterocycle functionalization, and drug development, making it a valuable intermediate in both medicinal and synthetic chemistry applications. References 2024. A kind of synthetic method of dasatinib intermediate. CN-118994050-A. URL: https://patents.google.com/patent/CN118994050A/en 2023. Thiazole compounds and methods of use thereof. US-2025145606-A1. URL: https://patents.google.com/patent/US2025145606A1/en |
| Market Analysis Reports |