Online Database of Chemicals from Around the World
| Zibo Feitian International Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.feitianindustry.com | |||
 | +86 (533) 276-3996 | |||
 | info@feitianindustry.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Premium supplier since 2022 | ||||
| Classification | API >> Special medicine >> Surgical medication |
|---|---|
| Name | Paraformaldehyde |
| Synonyms | p-Formaldehyde; Polyoxymethylene |
| Molecular Structure | 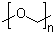 |
| Molecular Formula | (CH2O)n |
| CAS Registry Number | 30525-89-4 |
| EC Number | 690-727-5 |
| Density | 0.88 |
|---|---|
| Melting point | 120-170 °C |
| Flash point | 71 °C |
| Water solubility | sparingly soluble |
| Hazard Symbols |     GHS02;GHS05;GHS07;GHS08 Danger Details GHS02;GHS05;GHS07;GHS08 Danger Details |
|---|---|
| Risk Statements | H228-H302-H312-H315-H317-H318-H319-H332-H334-H335-H341-H351 Details |
| Safety Statements | P203-P210-P233-P240-P241-P260-P261-P264-P264+P265-P270-P271-P272-P280-P284-P301+P317-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P317-P318-P319-P321-P330-P332+P317-P333+P317-P337+P317-P342+P316-P362+P364-P370+P378-P403-P403+P233-P405-P501 Details |
| Transport Information | UN 2213 |
| SDS | Available |
|
Paraformaldehyde is a polymeric form of formaldehyde, a simple aldehyde with the chemical formula (CH2O)n, where "n" represents the number of repeating units. It typically appears as a white, powdery solid or a colorless granule and is primarily used as a source of formaldehyde in various chemical reactions and industrial applications. The discovery of paraformaldehyde can be traced back to the early 20th century, shortly after formaldehyde itself was first synthesized in 1859 by the German chemist August Wilhelm von Hofmann. Over time, paraformaldehyde emerged as a convenient solid form of formaldehyde, facilitating its use in laboratory and industrial settings. The polymerization of formaldehyde into paraformaldehyde occurs under certain conditions, such as low temperatures or high concentrations of formaldehyde. This reaction leads to a white, waxy solid that is soluble in water, making it an important compound in various fields of chemistry and industry. Paraformaldehyde is produced on a large scale through the depolymerization of formaldehyde in the presence of heat, and it can be readily converted back to formaldehyde when dissolved in water or treated with an acid. One of the primary applications of paraformaldehyde is as a source of formaldehyde in the production of resins and plastics. It is commonly used in the manufacturing of urea-formaldehyde and phenol-formaldehyde resins, which are widely utilized in the production of particleboard, plywood, and other wood products. These resins impart desirable properties such as durability, moisture resistance, and adhesion, making them essential in the construction and furniture industries. In addition to its role in resin production, paraformaldehyde is extensively used in laboratory settings as a fixative in histology and microscopy. It preserves biological tissues and cellular structures, allowing for better visualization and analysis under a microscope. Paraformaldehyde solutions are often preferred over formaldehyde solutions due to their lower toxicity and improved handling characteristics, making them suitable for use in various biological and medical research applications. Paraformaldehyde is also employed in the synthesis of various chemicals and pharmaceuticals. It serves as a reagent in organic synthesis, facilitating the formation of a wide range of compounds, including methylene-linked polymers, cyclic compounds, and more complex organic molecules. Researchers have exploited its reactivity to develop new synthetic pathways and methodologies in organic chemistry. Despite its widespread use, handling paraformaldehyde requires caution due to its potential health risks. Formaldehyde is a known irritant and has been classified as a possible human carcinogen. Therefore, appropriate safety measures must be implemented when working with paraformaldehyde in industrial and laboratory settings, including the use of personal protective equipment and adequate ventilation. In summary, paraformaldehyde is a vital chemical compound with diverse applications across various industries, particularly in the production of resins, laboratory fixatives, and organic synthesis. Its discovery and subsequent development have significantly contributed to advancements in material science and biological research, making it an essential compound in modern chemistry. References 1979. Catecholamines in the yolk sac epithelium of the rat. Brain Structure and Function, 159(1). DOI: 10.1007/bf00300013 1979. A cytochemical and immunofluorescence study of endocrine cells in the gut of the ascidian Styela clava. Cell and Tissue Research, 199(2). DOI: 10.1007/bf00237733 1979. Characterization of protein components of poly(A)-containing messenger ribonucleoproteins from cryptobiotic gastrulae of Artemia salina. Molecular Biology Reports, 5(1-2). DOI: 10.1007/bf00777490 |
| Market Analysis Reports |