Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Organic raw materials >> Amino compound >> Amide compound |
|---|---|
| Name | Paraherquamide |
| Synonyms | (1'S,6'R,7'R,8R,9'S)-6'-hydroxy-4,4,6',10',10',13'-hexamethylspiro[10H-[1,4]dioxepino[2,3-g]indole-8,11'-3,13-diazatetracyclo[5.5.2.01,9.03,7]tetradecane]-9,14'-dione |
| Molecular Structure | 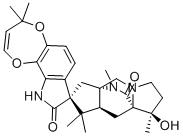 |
| Molecular Formula | C28H35N3O5 |
| Molecular Weight | 493.59 |
| CAS Registry Number | 77392-58-6 |
| SMILES | C[C@]1(CCN2[C@]13C[C@@H]4[C@](C2)(C[C@@]5(C4(C)C)C6=C(C7=C(C=C6)OC(C=CO7)(C)C)NC5=O)N(C3=O)C)O |
| Solubility | 5.804 mg/L (25 °C water) |
|---|---|
| Density | 1.4±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.674, Calc.* |
| Melting point | 290.08 °C |
| Boiling Point | 665.87 °C, 682.8±55.0 °C (760 mmHg), Calc.* |
| Flash Point | 366.7±31.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS06 Danger Details GHS06 Danger Details |
|---|---|
| Risk Statements | H300 Details |
| Safety Statements | P264-P270-P301+P310-P321-P330-P405-P501 Details |
| SDS | Available |
|
Paraherquamide is a naturally occurring alkaloid isolated from the fermentation products of the bacterium *Streptomyces** species*. This compound is part of a broader class of substances known for their bioactive properties, particularly in their ability to interact with biological systems in complex ways. Paraherquamide was first identified due to its potent pharmacological effects, and its structure and properties have since been the subject of extensive research. The discovery of paraherquamide marked an important milestone in natural product chemistry, as it presented unique molecular features that set it apart from other known alkaloids. Specifically, it contains a distinct polycyclic structure that includes an indole ring system fused with a piperazine ring, which is crucial for its biological activity. The first synthesis of paraherquamide was a significant achievement in the field, as it allowed researchers to study the compound in more detail, facilitating the exploration of its mechanism of action. In terms of its applications, paraherquamide has garnered attention for its potential therapeutic effects. It has been primarily researched for its ability to modulate neurotransmitter systems, with particular emphasis on its action as a selective serotonin receptor agonist. This suggests that paraherquamide could have applications in the treatment of various disorders related to neurotransmitter imbalances, including mood disorders and other psychiatric conditions. Moreover, studies have also examined its effect on other receptors, expanding the potential therapeutic scope. Additionally, paraherquamide’s antimicrobial properties have been explored, with some studies suggesting its utility in the treatment of infections caused by resistant strains of bacteria. Its unique structure and activity profile make it an attractive candidate for further development in the field of medicinal chemistry. The compound’s potential to serve as a template for the design of novel pharmaceuticals has been acknowledged by researchers seeking to leverage its bioactive properties for drug development. In conclusion, paraherquamide remains an intriguing natural product with a diverse range of applications, particularly in the fields of pharmacology and medicinal chemistry. Its discovery has contributed to a greater understanding of natural alkaloids and their potential for therapeutic use, providing a foundation for future drug development efforts aimed at addressing a variety of medical conditions. References 1990. Novel antinematodal and antiparasitic agents from Penicillium charlesii. I. Fermentation, isolation and biological activity. The Journal of Antibiotics, 43(11). DOI: 10.7164/antibiotics.43.1375 2003. Asymmetric, Stereocontrolled Total Synthesis of Paraherquamide A. Journal of the American Chemical Society, 125(36). DOI: 10.1021/ja036713+ 2023. Determinants of Subtype-Selectivity of the Anthelmintic Paraherquamide A on Caenorhabditis elegans Nicotinic Acetylcholine Receptors. Molecular Pharmacology, 103(6). DOI: 10.1124/molpharm.122.000601 |
| Market Analysis Reports |