Online Database of Chemicals from Around the World
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| West Bengal Chemical Industries Limited | India | |||
|---|---|---|---|---|
 | www.wbcil.com | |||
 | 08240399360 | |||
 | wbcilpvtltd@gmail.com | |||
| Chemical manufacturer since 1970 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic copper |
|---|---|
| Name | Cupric propionate |
| Synonyms | Copper(II) propanoate; Copper(II) propionate; Cupric propanoate |
| Molecular Structure | 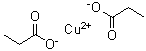 |
| Molecular Formula | Cu(C3H5O2)2 |
| Molecular Weight | 209.69 |
| CAS Registry Number | 3112-74-1 |
| SMILES | CCC(=O)[O-].CCC(=O)[O-].[Cu+2] |
| Hazard Symbols |    GHS05;GHS07;GHS09 Danger Details GHS05;GHS07;GHS09 Danger Details |
|---|---|
| Risk Statements | H302-H314-H400-H411 Details |
| Safety Statements | P260-P264-P270-P273-P280-P301+P312+P330-P301+P330+P331-P303+P361+P353-P304+P340+P310-P305+P351+P338+P310-P363-P391-P405-P501 Details |
| Transport Information | UN 1759 |
| SDS | Available |
|
Cupric propionate is an inorganic–organic coordination compound in which copper is present in the +2 oxidation state and coordinated to propionate ligands derived from propionic acid. Its empirical formula is generally represented as Cu(C2H5COO)2, and it is typically encountered as a blue-green crystalline solid, reflecting the characteristic color of hydrated Cu(II) complexes. The material is sparingly soluble in water but more soluble in certain organic solvents, and it can form hydrates depending on preparation and storage conditions. The preparation of cupric propionate is commonly achieved by the reaction of copper(II) oxide, copper(II) hydroxide, or copper(II) carbonate with propionic acid under controlled conditions. The process can be conducted in aqueous or alcoholic media, and subsequent evaporation or crystallization yields the product. Alternatively, metathesis reactions between soluble copper salts such as copper(II) sulfate and propionate salts like sodium or potassium propionate can also be used. Cupric propionate has been studied for applications where both the copper ion and the propionate ligand contribute functional properties. Copper, as an essential trace element, is involved in numerous enzymatic processes, and copper(II) salts can serve as nutritional supplements in animal feed when administered in appropriate doses. In this context, cupric propionate has been investigated as a bioavailable form of copper for livestock, offering a balance between solubility and stability. However, its use in animal nutrition must adhere to regulatory limits to avoid copper toxicity. In industrial chemistry, cupric propionate can function as a catalyst or catalyst precursor in organic synthesis, particularly in oxidation and coupling reactions where Cu(II) participates in redox cycles. Its moderate solubility and organic ligand environment may influence reaction rates and selectivity compared to more common copper salts such as cupric acetate or cupric chloride. The compound also exhibits antimicrobial properties due to the biocidal activity of copper ions, which can disrupt microbial membranes and enzyme systems. As a result, cupric propionate may find use in specialized biocidal formulations for coatings, textiles, or polymeric materials, where slow release of copper provides prolonged protection against microbial growth. This antimicrobial action, however, must be balanced against potential environmental accumulation of copper. While cupric propionate is chemically stable under ambient conditions, it should be stored in dry, airtight containers to prevent hydrolysis or reaction with atmospheric moisture and carbon dioxide, which can alter its composition. Direct contact with strong acids or bases should be avoided to prevent decomposition or ligand exchange reactions. Toxicological considerations are similar to those of other soluble copper(II) salts: ingestion of excessive amounts can lead to gastrointestinal irritation and systemic copper toxicity. Handling should be performed with appropriate protective equipment to avoid skin or eye contact, and disposal should comply with environmental regulations governing copper-containing substances. References 2024. Multifunctional vascular stent for releasing dual therapeutic gases and preparation method thereof. Publication Number: CN-119345488-A. Priority Date: 2024-11-01. 2024. Metal ion polyphenol coating material, preparation method and application thereof. Publication Number: CN-118976161-A. Priority Date: 2024-08-12. 2023. Metal-based anti-microbial additives and promoters for polymer compositions. Publication Number: EP-4458151-A1. Priority Date: 2023-10-02. |
| Market Analysis Reports |