Online Database of Chemicals from Around the World
| Choriway International Trade Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.choriway.com | |||
 | +86 (510) 8260-6408 | |||
 | +86 (510) 8270-0244 | |||
 | sales@choriway.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| TongVo Chemicals (Hangzhou) Limited | China | |||
|---|---|---|---|---|
 | www.tongvo.cn | |||
 | +86 (571) 8695-6296 | |||
 | +86 (571) 8790-8611 | |||
 | sales@tongvo.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| LTW (Jiangsu) LLC | China | |||
|---|---|---|---|---|
 | www.ltwfood.com | |||
 | +86 18505187090 | |||
 | sales9@ltwfood.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Choice Organochem LLP | India | |||
|---|---|---|---|---|
 | choiceorg.com | |||
 | +91 9618333877 | |||
 | web@choiceorg.com | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Celtic Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.celticchemicals.co.uk | |||
 | +44 (1656) 749-358 | |||
 | +44 (1656) 746-490 | |||
 | sales@celticchemicals.co.uk | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal sulfides and sulfates |
|---|---|
| Name | Cupric sulfate |
| Synonyms | Copper(II) sulfate |
| Molecular Structure | 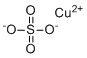 |
| Molecular Formula | CuSO4 |
| Molecular Weight | 159.60 |
| CAS Registry Number | 7758-98-7 |
| EC Number | 231-847-6 |
| SMILES | [O-]S(=O)(=O)[O-].[Cu+2] |
| Density | 3.603 g/mL (20 °C) |
|---|---|
| Melting point | 560 °C (dec.) |
| Water solubility | 14.3% w/w (20 °C) |
| Hazard Symbols |    GHS05;GHS07;GHS09 Danger Details GHS05;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H318-H319-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P273-P280-P301+P317-P302+P352-P305+P351+P338-P305+P354+P338-P317-P321-P330-P332+P317-P337+P317-P362+P364-P391-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 3077; UN 9109 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Copper sulfate, also known as copper (II) sulfate or CuSO4, was first discovered and isolated in the 17th century by German-Dutch chemist Johann Rudolf Glauber. It consists of copper (Cu²?) ions and sulfate (SO4²?) ions, forming blue crystals that are soluble in water. The compound exists in a variety of forms, including anhydrous (CuSO4) and hydrated forms, such as copper sulfate pentahydrate (CuSO4·5H2O), which is the most common and stable form. Copper sulfate is widely used in agriculture as a fungicide and insecticide. It helps control fungal diseases in crops such as grapes, potatoes, and tomatoes by inhibiting the germination and growth of fungal spores. In addition, it can be used as a molluscicide to control snail and slug populations in agricultural fields. In industrial applications, copper sulfate is used in the electroplating process to deposit copper onto metal surfaces. This electroplating method enhances the conductivity, corrosion resistance, and aesthetics of metals used in electronics, automotive parts, and decorative items. Copper sulfate is used in the production of pigments and dyes, especially for making blue and green pigments. It is used as a mordant in dyeing textiles and as a pigment in paints and ceramics, imparting bright and stable colors. In the laboratory, copper sulfate is used as a standard chemical reagent for a variety of analytical and qualitative tests. Its unique blue color makes it useful for identifying chemical reactions and the presence of other substances in solution. Copper sulfate is used to demonstrate chemical principles, including precipitation reactions, the hydration state of salts, and crystal growth. Its availability and clear crystal structure make it an ideal substance for chemical experiments. Historically, copper sulfate has been used in medicine as an emetic (to induce vomiting) and to treat copper deficiency. However, its medical use is limited today due to potential toxicity and the emergence of safer alternatives. References 2003. Susceptibility and features of the ultrastructure of Prototheca zopfii following exposure to copper sulphate, silver nitrate and chlorexidine. Mycopathologia, 156(1). DOI: 10.1023/a:1021313118632 2007. Development of a daphnia magna DNA microarray for evaluating the toxicity of environmental chemicals. Environmental Toxicology and Chemistry, 26(4). DOI: 10.1897/06-075r.1 1987. The effect of a combination of copper and hexetidine on plaque formation and the amount of copper retained by dental plaque bacteria. Acta Odontologica Scandinavica, 45(6). DOI: 10.3109/00016358709096368 |
| Market Analysis Reports |