Online Database of Chemicals from Around the World
| Anhui Modern Chem-Pharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amcpc.cn | |||
 | +86 (554) 361-1480 | |||
 | +86 (554) 361-1480 | |||
 | amcp@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shanghai Nanxiang Reagent Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nx-reagent.com | |||
 | +86 (21) 3912-7448 +86 13512139665 | |||
 | +86 (21) 3912-7431 | |||
 | xumiao@nx-reagent.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Kingchem Inc. | USA | |||
|---|---|---|---|---|
 | www.kingchem.com | |||
 | +1 (800) 211-4330 (201) 825-9988 | |||
 | +1 (201) 825-9148 | |||
 | customer-service@kingchem.com | |||
| Chemical manufacturer since 1994 | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic acid |
|---|---|
| Name | 5-Chlorosalicylic acid |
| Synonyms | 5-Chloro-2-hydroxybenzoic acid |
| Molecular Structure | 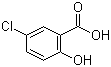 |
| Molecular Formula | C7H5ClO3 |
| Molecular Weight | 172.57 |
| CAS Registry Number | 321-14-2 |
| EC Number | 206-283-9 |
| SMILES | C1=CC(=C(C=C1Cl)C(=O)O)O |
| Melting point | 171-176 °C |
|---|---|
| Water solubility | 1 g/L (20 °C) |
| Density | 1.5±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.630, Calc.* |
| Boiling Point | 320.5±27.0 °C (760 mmHg), Calc.* |
| Flash Point | 147.6±23.7 °C, Calc.* |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P273-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
5-Chlorosalicylic acid, a chlorinated derivative of salicylic acid, is an important compound in organic chemistry and pharmaceutical applications. Its discovery dates back to the early 20th century, when researchers began exploring the properties and reactions of salicylic acid and its derivatives. Salicylic acid itself was first isolated from willow bark and has a long history of use, particularly in the development of aspirin. The introduction of chlorine atoms into the salicylic acid structure produced 5-chlorosalicylic acid, which possesses unique chemical and biological properties. The synthesis of 5-chlorosalicylic acid typically involves the chlorination of salicylic acid. Various methods have been developed to achieve this, including electrophilic aromatic substitution reactions, where chlorine is introduced at the 5-position of the aromatic ring. This modification not only enhances the compound's solubility in organic solvents but also imparts specific pharmacological activities. 5-Chlorosalicylic acid is primarily recognized for its role as a pharmaceutical intermediate. It has been used in the synthesis of various therapeutic agents, particularly in the field of anti-inflammatory and analgesic medications. Its structural similarity to salicylic acid allows it to exhibit similar biological activities, including anti-inflammatory properties. Researchers have also explored its potential as an antimicrobial agent, showcasing its efficacy against a range of bacterial and fungal strains. In addition to its pharmaceutical applications, 5-chlorosalicylic acid serves as a versatile reagent in organic synthesis. It is utilized in the production of agrochemicals, dyes, and other fine chemicals. Its chlorinated nature enhances the compound's reactivity, making it a valuable building block in the synthesis of more complex molecules. Moreover, 5-chlorosalicylic acid has been studied for its potential role in cancer research. Preliminary studies suggest that it may inhibit the growth of certain cancer cell lines, indicating a possible application in cancer therapy. These findings have prompted further investigation into its mechanism of action and potential therapeutic benefits. In summary, 5-chlorosalicylic acid is a significant chemical compound with a rich history and diverse applications. Its discovery marked a notable advancement in organic chemistry, and its continued exploration in pharmaceuticals and agrochemicals highlights its importance in contemporary research and industry. The compound's unique properties and potential therapeutic applications make it a subject of ongoing scientific interest, paving the way for future discoveries and innovations. References 2022. Insights on structure and interactions of 2-amino-4-methoxy-6-methylpyrimidinium salts with 4-aminosalicylate and 5-chlorosalicylate: a combined experimental and theoretical charge-density analysis. Acta Crystallographica. Section C, Structural Chemistry, 78(2). DOI: 10.1107/s2053229622001280 2015. The antioxidant properties of salicylate derivatives: A possible new mechanism of anti-inflammatory activity. Bioorganic & Medicinal Chemistry Letters, 25(21). DOI: 10.1016/j.bmcl.2015.07.001 2015. Design and synthesis of 5-chloro-2-hydroxy-3-triazolylbenzoic acids as HIV integrase inhibitors. Medicinal Chemistry Research, 24(6). DOI: 10.1007/s00044-015-1325-9 |
| Market Analysis Reports |