Online Database of Chemicals from Around the World
| Shanghai Boka-chem Tech Inc. | China | |||
|---|---|---|---|---|
 | www.bokachem.com | |||
 | +86 (21) 6478-0600 | |||
 | +86 (21) 6459-2060 | |||
 | lidong@bokachem.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2009 | ||||
| Sichuan Zhongbang Technical Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhongbangst.com | |||
 | +86 (830) 258-5019 +86 18982761636 | |||
 | +86 (830) 258-9033 | |||
 | sales@zhongbangst.com sheldon0chen@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BeiJing VsciChem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.vscichem.com | |||
 | +86 (10) 5248-3976 | |||
 | +86 (10) 6027-0130 | |||
 | vscichem@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhengzhou Changkuan Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.changkuantech.com | |||
 | +86 (371) 6376-9919 +86 18530983798 | |||
 | +86 (371) 6360-3986 | |||
 | sales5@changkuantech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Xinxiang Runyu Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runvmat.com | |||
 | +86 (373) 775-9608 | |||
 | +86 (373) 775-9608 | |||
 | sales@runvmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | Butyldi-1-adamantylphosphine |
| Synonyms | bis(1-adamantyl)-butylphosphane |
| Molecular Structure | 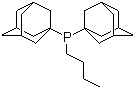 |
| Molecular Formula | C24H39P |
| Molecular Weight | 358.55 |
| CAS Registry Number | 321921-71-5 |
| EC Number | 691-708-4 |
| SMILES | CCCCP(C12CC3CC(C1)CC(C3)C2)C45CC6CC(C4)CC(C6)C5 |
| Melting point | 100 °C (Expl.) |
|---|---|
| Boiling Point | 449.6±12.0 °C (760 mmHg), Calc.* |
| Flash Point | 239.0±25.8 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Butyldi-1-adamantylphosphine is an organophosphorus compound that plays a significant role in the field of organometallic chemistry and catalysis. It is a derivative of adamantane, a polycyclic hydrocarbon, where one of the hydrogen atoms is replaced by a butyl group, and the phosphorus atom is attached to the adamantyl group. This unique structure imparts both steric and electronic properties that are valuable in various chemical applications. The compound has been used primarily as a ligand in metal-catalyzed reactions, benefiting from the stability and steric protection offered by the adamantyl group. The discovery of butyldi-1-adamantylphosphine can be traced back to the ongoing research on phosphines in the late 20th century, which focused on developing sterically demanding ligands to control the reactivity and selectivity of transition metal catalysts. Researchers identified that phosphines with bulky groups, such as adamantyl, could provide enhanced stability to metal-ligand complexes and improve their performance in certain catalytic processes. This led to the development of butyldi-1-adamantylphosphine as a ligand for use in metal-catalyzed transformations, where its unique steric environment could help achieve high selectivity. The synthesis of butyldi-1-adamantylphosphine typically involves the reaction of 1-adamantyl lithium with a chlorophosphine precursor, followed by substitution to introduce the butyl group. This method ensures that the adamantyl group remains intact while the butyl group is successfully attached to the phosphorus atom. The resulting phosphine ligand is stable and can be purified through standard organic chemistry techniques, such as chromatography. One of the primary applications of butyldi-1-adamantylphosphine is in catalytic processes, particularly those involving transition metals like palladium, rhodium, and platinum. The bulkiness of the adamantyl group in the ligand helps to create a more rigid coordination environment, which can lead to increased selectivity and reactivity in reactions such as cross-coupling, hydrogenation, and C-H activation. These reactions are crucial in the synthesis of fine chemicals, pharmaceuticals, and agrochemicals. The phosphine ligand is also used in reactions that require enhanced stability and control over the catalyst's performance. Butyldi-1-adamantylphosphine has found applications in the synthesis of complex organic molecules, where precise control over the stereochemistry and reactivity of the intermediates is essential. The ligand’s ability to improve the efficiency of metal-catalyzed reactions has made it a valuable tool in both academic research and industrial processes. References 2017. Tri(1-adamantyl)phosphine: Exceptional Catalytic Effects Enabled by the Synergy of Chemical Stability, Donicity, and Polarizability. Synlett, 28(4). DOI: 10.1055/s-0036-1588128 2017. Palladium-catalysed synthesis of triaryl(heteroaryl)methanes. Nature Communications, 8(1). DOI: 10.1038/ncomms14641 2017. Synthesis and preliminary PET imaging of 11C and 18F isotopologues of the ROS1/ALK inhibitor lorlatinib. Nature Communications, 8(1). DOI: 10.1038/ncomms15761 |
| Market Analysis Reports |